International prognostic scoring system for mastocytosis (IPSM): a retrospective cohort study
- PMID: 31676322
- PMCID: PMC7115823
- DOI: 10.1016/S2352-3026(19)30166-8
International prognostic scoring system for mastocytosis (IPSM): a retrospective cohort study
Abstract
Background: The WHO classification separates mastocytosis into distinct variants, but prognostication remains a clinical challenge. The aim of this study was to improve prognostication for patients with mastocytosis.
Methods: We analysed data of the registry of the European Competence Network on Mastocytosis including 1639 patients (age 17-90 years) diagnosed with mastocytosis according to WHO criteria between Jan 12, 1978, and March 16, 2017. Univariate and multivariate analyses with Cox regression were applied to identify prognostic variables predicting survival outcomes and to establish a prognostic score. We validated this International Prognostic Scoring System in Mastocytosis (IPSM) with data of 462 patients (age 17-79 years) from the Spanish network Red Española de Mastocitosis diagnosed between Jan 22, 1998, and Nov 2, 2017.
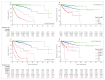
Findings: The prognostic value of the WHO classification was confirmed in our study (p<0·0001). For patients with non-advanced mastocytosis (n=1380), we identified age 60 years or older (HR 10·75, 95% CI 5·68-20·32) and a concentration of alkaline phosphatase 100 U/L or higher (2·91, 1·60-5·30) as additional independent prognostic variables for overall survival. The resulting scoring system divided patients with non-advanced mastocytosis into three groups: low (no risk factors), intermediate 1 (one risk factor), and intermediate 2 (two risk factors). Overall survival and progression-free survival differed significantly among these groups (p<0·0001). In patients with advanced mastocytosis (n=259), age 60 years or older (HR 2·14, 95% CI 1·42-3·22), a concentration of tryptase 125 ng/mL or higher (1·81, 1·20-2·75), a leukocyte count of 16 × 109 per L or higher (1·88, 1·27-2·79), haemoglobin of 11 g/dL or lower (1·71, 1·13-2·57), a platelet count of 100 × 109 per L or lower (1·63, 1·13-2·34), and skin involvement (0·46, 0·30-0·69) were prognostic variables. Based on these variables, a separate score for advanced mastocytosis with four risk categories was established, with significantly different outcomes for overall survival and progression-free survival (p<0·0001). The prognostic value of both scores was confirmed in 413 patients with non-advanced disease and 49 with advanced mastocytosis from the validation cohort.
Interpretation: The IPSM scores for patients with non-advanced and advanced mastocytosis can be used to predict survival outcomes and guide treatment decisions. However, the predictive value of the IPSM needs to be confirmed in forthcoming trials.
Funding: Austrian Science Fund, Deutsche Forschungsgemeinschaft, Koeln Fortune Program, Charles and Ann Johnson Foundation, Instituto de Salud Carlos III, Fondos FEDER, Research-Foundation Flanders/Fonds Wetenschappelijk Onderzoek, Clinical Research-Fund of the University Hospitals Leuven, and Research-Foundation Flanders/Fonds Wetenschappelijk Onderzoek.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
WRS declares honoraria from AbbVie, Amgen, Celgene, Daiichi Sankyo, Deciphera, Incyte, Jazz, Novartis, Pfizer, and Thermo Fisher; and travel grants from Pfizer and Roche. BvA, IA-T, and ASY declare honoraria from Novartis. KVG declares honoraria from Pfizer, Novartis, Roche, BMS, Sanofi, and Incyte; and travel grants from Roche and AbbVie. RZ declares honoraria from Deciphera, Novartis, and Takeda. MB declares honoraria from Pfizer, Amgen, and Incyte; and research funding from Novartis. CE declares honoraria from Novartis and Pfizer. KS declares honoraria from Novartis; and travel grants from AbbVie. NvB declares honoraria from AstraZeneca, Amgen, Novartis, and BMS; and research funding from Novartis. AZ declares honoraria from or has participated in trials for AbbVie, Almirall, Beiersdorf Dermo Medical, Bencard Allergie, BMS, Celgene, Eli Lilly, GSK, Janssen-Cilag, Miltenyi Biotec, Novartis, Sanofi-Aventis, and Takeda Pharma. DF declares honoraria from Novartis, Pfizer, and Roche; and travel grants from Roche. JP declares honoraria from Exion, BMS, Boehringer Ingelheim, Grünenthal, MSD, Novartis, Pfizer, Chugai, and Roche. DN declares research funding from Novartis; and non-financial support from Amgen and Cellectis. CB declares honoraria from Thermo Fisher Phadia; and travel grants from Shire/Takeda, HAL, and ALK. KH declares honoraria from ALK, Blueprint, Deciphera, and Novartis; and research funding from Euroimmun. MT declares honoraria from Blueprint, Deciphera, and Novartis. AR declares honoraria from Blueprint, Deciphera, Incyte, and Novartis. AO declares honoraria from Becton Dickinson, Janssen, and Novartis; research funding from Becton Dickinson, Cytognos, and Immunostep; and is an inventor on patents licensed to Cytognos, Immunostep, Fagotrace, and Becton-Dickinson that return royalties to the University of Salamanca and EuroFlow. OH declares honoraria from ABscience; and research funding from Alexion, Celgene, and Novartis. JG declares honoraria and research funding from Blueprint, Deciphera, and Novartis. MA declares honoraria from Deciphera, Allakos, and Blueprint; and research funding from Allakos and Blueprint. HCK-N declares research funding from Novartis. PV declares honoraria from Blueprint, Celgene, Deciphera, Incyte, Novartis, and Pfizer; and research funding from Pfizer, Incyte, and Celgene. MK, JNGOE, AG, MN, EH, PB, CP, AI, SM, RP, MJ, ABF, FC, KB, AJK, MD, HH, VS, AB, and BN declare no competing interests.
Figures

Comment in
-
A useful add-on international prognostic score for mastocytosis?Lancet Haematol. 2019 Dec;6(12):e604-e605. doi: 10.1016/S2352-3026(19)30212-1. Epub 2019 Oct 31. Lancet Haematol. 2019. PMID: 31676323 No abstract available.
References
-
- Cohen SS, Skovbo S, Vestergaard H, et al. Epidemiology of systemic mastocytosis in Denmark. Br J Haematol. 2014;166:521–28. - PubMed
-
- Valent P, Horny HP, Escribano L, et al. Diagnostic criteria and classification of mastocytosis: a consensus proposal. Leuk Res. 2001;25:603–25. - PubMed
-
- Lim KH, Tefferi A, Lasho TL, et al. Systemic mastocytosis in 342 consecutive adults: survival studies and prognostic factors. Blood. 2009;113:5727–36. - PubMed
-
- Valent P, Akin C, Escribano L, et al. Standards and standardization in mastocytosis: consensus statements on diagnostics, treatment recommendations and response criteria. Eur J Clin Invest. 2007;37:435–53. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

