Immune biology of glioma-associated macrophages and microglia: functional and therapeutic implications
- PMID: 31679017
- PMCID: PMC7442334
- DOI: 10.1093/neuonc/noz212
Immune biology of glioma-associated macrophages and microglia: functional and therapeutic implications
Abstract
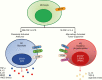
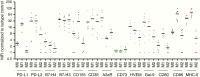
CNS immune defenses are marshaled and dominated by brain resident macrophages and microglia, which are the innate immune sentinels and frontline host immune barriers against various pathogenic insults. These myeloid lineage cells are the predominant immune population in gliomas and can constitute up to 30-50% of the total cellular composition. Parenchymal microglial cells and recruited monocyte-derived macrophages from the periphery exhibit disease-specific phenotypic characteristics with spatial and temporal distinctions and are heterogeneous subpopulations based on their molecular signatures. A preponderance of myeloid over lymphoid lineage cells during CNS inflammation, including gliomas, is a contrasting feature of brain immunity relative to peripheral immunity. Herein we discuss glioma-associated macrophage and microglia immune biology in the context of their identity, molecular drivers of recruitment, nomenclature and functional paradoxes, therapeutic reprogramming and polarization strategies, relevant challenges, and our perspectives on therapeutic modulation.
Keywords: glioma; immune suppression; macrophages; microglia.
© The Author(s) 2019. Published by Oxford University Press on behalf of the Society for Neuro-Oncology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

