Enzymatically Functionalized Composite Materials Based on Nanocellulose and Poly(Vinyl Alcohol) Cryogel and Possessing Antimicrobial Activity
- PMID: 31689944
- PMCID: PMC6862455
- DOI: 10.3390/ma12213619
Enzymatically Functionalized Composite Materials Based on Nanocellulose and Poly(Vinyl Alcohol) Cryogel and Possessing Antimicrobial Activity
Abstract
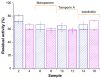
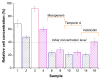
In the present work, innovative composite biomaterials possessing bactericidal properties and based on the hexahistidine-tagged organophosphorus hydrolase (His6-OPH) entrapped in the poly(vinyl alcohol) cryogel (PVA-CG)/bacterial cellulose (BC) were developed. His6-OPH possesses lactonase activity, with a number of N-acyl homoserine lactones being the inducers of Gram-negative bacterial resistance. The enzyme can also be combined with various antimicrobial agents (antibiotics and antimicrobial peptides) to improve the efficiency of their action. In this study, such an effect was shown for composite biomaterials when His6-OPH was entrapped in PVA-CG/BC together with β-lactam antibiotic meropenem or antimicrobial peptides temporin A and indolicidin. The residual catalytic activity of immobilized His6-OPH was 60% or more in all the composite samples. In addition, the presence of BC filler in the PVA-CG composite resulted in a considerable increase in the mechanical strength and heat endurance of the polymeric carrier compared to the BC-free cryogel matrix. Such enzyme-containing composites could be interesting in the biomedical field to help overcome the problem of antibiotic resistance of pathogenic microorganisms.
Keywords: antimicrobial peptides; bacterial cellulose; bactericidal activity; immobilized hexahistidine-tagged organophosphorus hydrolase; poly(vinyl alcohol) cryogel; β-lactam antibiotic.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Defoirdt T., Boon N., Bossier P., Verstraete W. Disruption of bacterial quorum sensing: An unexplored strategy to fight infections in aquaculture. Aquaculture. 2004;240:69–88. doi: 10.1016/j.aquaculture.2004.06.031. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

