Gold nanoparticle-adjuvanted S protein induces a strong antigen-specific IgG response against severe acute respiratory syndrome-related coronavirus infection, but fails to induce protective antibodies and limit eosinophilic infiltration in lungs
- PMID: 31692019
- PMCID: PMC7168429
- DOI: 10.1111/1348-0421.12754
Gold nanoparticle-adjuvanted S protein induces a strong antigen-specific IgG response against severe acute respiratory syndrome-related coronavirus infection, but fails to induce protective antibodies and limit eosinophilic infiltration in lungs
Abstract
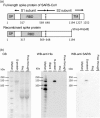
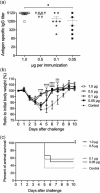
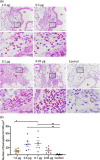
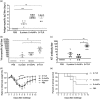
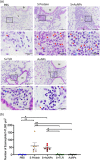
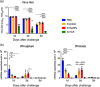
The spike (S) protein of coronavirus, which binds to cellular receptors and mediates membrane fusion for cell entry, is a candidate vaccine target for blocking coronavirus infection. However, some animal studies have suggested that inadequate immunization against severe acute respiratory syndrome coronavirus (SARS-CoV) induces a lung eosinophilic immunopathology upon infection. The present study evaluated two kinds of vaccine adjuvants for use with recombinant S protein: gold nanoparticles (AuNPs), which are expected to function as both an antigen carrier and an adjuvant in immunization; and Toll-like receptor (TLR) agonists, which have previously been shown to be an effective adjuvant in an ultraviolet-inactivated SARS-CoV vaccine. All the mice immunized with more than 0.5 µg S protein without adjuvant escaped from SARS after infection with mouse-adapted SARS-CoV; however, eosinophilic infiltrations were observed in the lungs of almost all the immunized mice. The AuNP-adjuvanted protein induced a strong IgG response but failed to improve vaccine efficacy or to reduce eosinophilic infiltration because of highly allergic inflammatory responses. Whereas similar virus titers were observed in the control animals and the animals immunized with S protein with or without AuNPs, Type 1 interferon and pro-inflammatory responses were moderate in the mice treated with S protein with and without AuNPs. On the other hand, the TLR agonist-adjuvanted vaccine induced highly protective antibodies without eosinophilic infiltrations, as well as Th1/17 cytokine responses. The findings of this study will support the development of vaccines against severe pneumonia-associated coronaviruses.
Keywords: adjuvant; coronavirus; eosinophils; gold nanoparticles; immunopathology; mouse model.
© 2019 The Societies and John Wiley & Sons Australia, Ltd.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures
Similar articles
-
Severe acute respiratory syndrome-associated coronavirus vaccines formulated with delta inulin adjuvants provide enhanced protection while ameliorating lung eosinophilic immunopathology.J Virol. 2015 Mar;89(6):2995-3007. doi: 10.1128/JVI.02980-14. Epub 2014 Dec 17. J Virol. 2015. PMID: 25520500 Free PMC article.
-
Effects of Toll-like receptor stimulation on eosinophilic infiltration in lungs of BALB/c mice immunized with UV-inactivated severe acute respiratory syndrome-related coronavirus vaccine.J Virol. 2014 Aug;88(15):8597-614. doi: 10.1128/JVI.00983-14. Epub 2014 May 21. J Virol. 2014. PMID: 24850731 Free PMC article.
-
A double-inactivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge.J Virol. 2011 Dec;85(23):12201-15. doi: 10.1128/JVI.06048-11. Epub 2011 Sep 21. J Virol. 2011. PMID: 21937658 Free PMC article.
-
Severe acute respiratory syndrome vaccine development: experiences of vaccination against avian infectious bronchitis coronavirus.Avian Pathol. 2003 Dec;32(6):567-82. doi: 10.1080/03079450310001621198. Avian Pathol. 2003. PMID: 14676007 Free PMC article. Review.
-
Protective Immunity against SARS Subunit Vaccine Candidates Based on Spike Protein: Lessons for Coronavirus Vaccine Development.J Immunol Res. 2020 Jul 18;2020:7201752. doi: 10.1155/2020/7201752. eCollection 2020. J Immunol Res. 2020. PMID: 32695833 Free PMC article. Review.
Cited by
-
Tracking SARS-CoV-2: Novel Trends and Diagnostic Strategies.Diagnostics (Basel). 2021 Oct 26;11(11):1981. doi: 10.3390/diagnostics11111981. Diagnostics (Basel). 2021. PMID: 34829328 Free PMC article. Review.
-
Identification of a Novel Neutralizing Epitope on the N-Terminal Domain of the Human Coronavirus 229E Spike Protein.J Virol. 2022 Feb 23;96(4):e0195521. doi: 10.1128/JVI.01955-21. Epub 2021 Dec 15. J Virol. 2022. PMID: 34908442 Free PMC article.
-
Potential therapeutic targets and promising drugs for combating SARS-CoV-2.Br J Pharmacol. 2020 Jul;177(14):3147-3161. doi: 10.1111/bph.15092. Epub 2020 Jun 5. Br J Pharmacol. 2020. PMID: 32368792 Free PMC article. Review.
-
Therapeutic approaches in COVID-19 followed before arrival of any vaccine.Mater Today Proc. 2022;48:1258-1264. doi: 10.1016/j.matpr.2021.08.265. Epub 2021 Sep 3. Mater Today Proc. 2022. PMID: 34493973 Free PMC article.
-
Preparation and immunological properties of a nanovaccine against Pseudomonas aeruginosa based on gold nanoparticles and detoxified lipopolysaccharide.Iran J Basic Med Sci. 2021 Feb;24(2):203-212. doi: 10.22038/ijbms.2020.50732.11550. Iran J Basic Med Sci. 2021. PMID: 33953860 Free PMC article. No abstract available.
References
-
- Droste C, Gunther S, Preiser W, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967‐76. - PubMed
-
- Ksiazek TG, Erdman D, Goldsmith CS, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953‐66. - PubMed
-
- Lee N, Hui D, Wu A, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986‐94. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

