Asymmetric Reduction of (R)-Carvone through a Thermostable and Organic-Solvent-Tolerant Ene-Reductase
- PMID: 31692216
- PMCID: PMC7216909
- DOI: 10.1002/cbic.201900599
Asymmetric Reduction of (R)-Carvone through a Thermostable and Organic-Solvent-Tolerant Ene-Reductase
Abstract
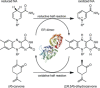
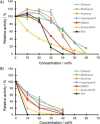
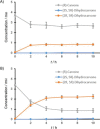
Ene-reductases allow regio- and stereoselective reduction of activated C=C double bonds at the expense of nicotinamide adenine dinucleotide cofactors [NAD(P)H]. Biological NAD(P)H can be replaced by synthetic mimics to facilitate enzyme screening and process optimization. The ene-reductase FOYE-1, originating from an acidophilic iron oxidizer, has been described as a promising candidate and is now being explored for applied biocatalysis. Biological and synthetic nicotinamide cofactors were evaluated to fuel FOYE-1 to produce valuable compounds. A maximum activity of (319.7±3.2) U mg-1 with NADPH or of (206.7±3.4) U mg-1 with 1-benzyl-1,4-dihydronicotinamide (BNAH) for the reduction of N-methylmaleimide was observed at 30 °C. Notably, BNAH was found to be a promising reductant but exhibits poor solubility in water. Different organic solvents were therefore assayed: FOYE-1 showed excellent performance in most systems with up to 20 vol% solvent and at temperatures up to 40 °C. Purification and application strategies were evaluated on a small scale to optimize the process. Finally, a 200 mL biotransformation of 750 mg (R)-carvone afforded 495 mg of (2R,5R)-dihydrocarvone (>95 % ee), demonstrating the simplicity of handling and application of FOYE-1.
Keywords: Old Yellow Enzymes; biocatalysis; biotransformations; cofactor mimics; oxidoreductases; solvent stability.
© 2019 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Conflict of interest statement
Figures

References
-
- None
-
- Alarcon J., Lamilla C., Cespedes C. L., Ind. Crop. Prod. 2013, 42, 268–272;
-
- Aubin Y., Audran G., Monti H., Tetrahedron Lett. 2006, 47, 3669–3671;
-
- de Rouville H. P. J., Vives G., Tur E., Rapenne G., Crassous J., New J. Chem. 2009, 33, 293–299;
-
- Dong Y., McCullough K. J., Wittlin S., Chollet J., Vennerstrom J. L., Bioorg. Med. Chem. Lett. 2010, 20, 6359–6361; - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

