Efficacy and Safety of Dupilumab in Adolescents With Uncontrolled Moderate to Severe Atopic Dermatitis: A Phase 3 Randomized Clinical Trial
- PMID: 31693077
- PMCID: PMC6865265
- DOI: 10.1001/jamadermatol.2019.3336
Efficacy and Safety of Dupilumab in Adolescents With Uncontrolled Moderate to Severe Atopic Dermatitis: A Phase 3 Randomized Clinical Trial
Abstract
Importance: Adolescents with atopic dermatitis (AD) have high disease burden negatively affecting quality of life, with limited treatment options. The efficacy and safety of dupilumab, a monoclonal antibody, approved for treatment in adolescent patients with inadequately controlled AD, remain unknown in this patient population.
Objective: To assess the efficacy and safety of dupilumab monotherapy in adolescents with moderate to severe inadequately controlled AD.
Design, setting, and participants: A randomized, double-blind, parallel-group, phase 3 clinical trial was conducted at 45 US and Canadian centers between March 21, 2017, and June 5, 2018. A total of 251 adolescents with moderate to severe AD inadequately controlled by topical medications or for whom topical therapy was inadvisable were included.
Interventions: Patients were randomized (1:1:1; interactive-response system; stratified by severity and body weight) to 16-week treatment with dupilumab, 200 mg (n = 43; baseline weight <60 kg), or dupilumab, 300 mg (n = 39; baseline weight ≥60 kg), every 2 weeks; dupilumab, 300 mg, every 4 weeks (n = 84); or placebo (n = 85).
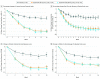
Main outcomes and measures: Proportion of patients with 75% or more improvement from baseline in Eczema Area and Severity Index (EASI-75) (scores range from 0 to 72, with higher scores indicating greater severity) and Investigator's Global Assessment (IGA) 0 or 1 on a 5-point scale (scores range from 0 to 4, with higher scores indicating greater severity) at week 16.
Results: A total of 251 patients were randomized (mean [SD] age, 14.5 [1.7] years; 148 [59.0%] male). Of 250 patients with data available on concurrent allergic conditions, most had comorbid type 2 diseases (asthma, 134 [53.6%]; food allergies, 60.8%; allergic rhinitis, 65.6%). A total of 240 patients (95.6%) completed the study. Dupilumab achieved both coprimary end points at week 16. The proportion of patients with EASI-75 improvement from baseline increased (every 2 weeks, 41.5%; every 4 weeks, 38.1%; placebo, 8.2%) with differences vs placebo of 33.2% (95% CI, 21.1%-45.4%) for every 2 weeks and 29.9% (95% CI, 17.9%-41.8%) for every 4 weeks (P < .001). Efficacy of the every-2-week regimen was generally superior to the every-4-week regimen. Patients in the dupilumab arms had higher percentage values of conjunctivitis (every 2 weeks, 9.8%; every 4 weeks, 10.8%; placebo, 4.7%) and injection-site reactions (every 2 weeks, 8.5%; every 4 weeks, 6.0%; placebo, 3.5%), and lower nonherpetic skin infections (every 2 weeks, 9.8%; every 4 weeks, 9.6%; placebo, 18.8%).
Conclusions and relevance: In this study, dupilumab significantly improved AD signs, symptoms, and quality of life in adolescents with moderate to severe AD, with an acceptable safety profile. Placebo-corrected efficacy and safety of dupilumab were similar in adolescents and adults.
Trial registration: ClinicalTrials.gov identifier: NCT03054428.
Conflict of interest statement
Figures


References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

