Cellular elements in the developing caecum of Japanese quail (Coturnix coturnix japonica): morphological, morphometrical, immunohistochemical and electron-microscopic studies
- PMID: 31700017
- PMCID: PMC6838329
- DOI: 10.1038/s41598-019-52335-x
Cellular elements in the developing caecum of Japanese quail (Coturnix coturnix japonica): morphological, morphometrical, immunohistochemical and electron-microscopic studies
Abstract
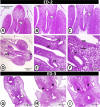
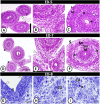
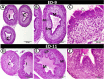
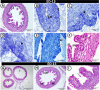
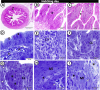
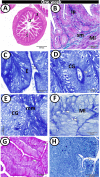
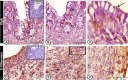
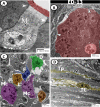
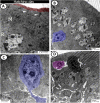
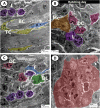
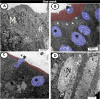
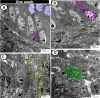
The present study aims to investigate the histological, histochemical and electron microscopic changes of the caecal proximal part of Japanese quail during both pre- and post-hatching periods starting from the 2nd embryonic day (ED) until four weeks post-hatching. On the 2nd and 3rd ED, the primordia of caeca appeared as bilateral swelling on the wall of the hindgut. On the 7th ED, the lamina propria/submucosa contained the primordia of glands. On the 8th ED, rodlet cells could be observed amongst the epithelial cells. On the 9th ED, the caeca began to divide into three parts with more developed layers. With age, the height and number of villi increased. On the 13th ED, immature microfold cells (M-cells) could be identified between the surface epithelium of the villi. The caecal tonsils (CTs) appeared in the form of aggregations of lymphocytes, macrophages, dendritic cells and different types of leukocytes. Telocytes and crypts of Lieberkuhn were observed at this age. On hatching day, the crypts of Lieberkuhn were well-defined and formed of low columnar epithelium, goblet cells, and enteroendocrine cells. Post-hatching, the lumen was filled with villi that exhibited two forms: (1) tongue-shaped villi with tonsils and (2) finger-shaped ones without tonsils. The villi lining epithelium contained simple columnar cells with microvilli that were dispersed with many goblet cells, in addition to the presence of a high number of intra-epithelial lymphocytes and basophils. Moreover, the submucosa was infiltrated by numerous immune cells. CD3 immunomarker was expressed in intraepithelial lymphocytes, while CD20 immunomarker showed focal positivity in CTs. In conclusion, the caecal immune structures of quails at post-hatching were more developed than those in pre-hatching life. The high frequency of immune cells suggests that this proximal part may be a site for immunological surveillance in the quail caecum. The cellular organisation of the caecum and its relation to the immunity was discussed.
Conflict of interest statement
The authors declare no competing interests.
Figures






Similar articles
-
Morphological Characteristics of the Developing Cecum of Japanese Quail (Coturnix coturnix japonica).Microsc Microanal. 2019 Aug;25(4):1017-1031. doi: 10.1017/S1431927619000655. Epub 2019 Jun 6. Microsc Microanal. 2019. PMID: 31169105
-
Developmental morpho-analysis of the caecum in Japanese quail embryos (Coturnix coturnix japonica).Microsc Res Tech. 2024 Nov;87(11):2540-2554. doi: 10.1002/jemt.24632. Epub 2024 Jun 12. Microsc Res Tech. 2024. PMID: 38864478
-
Histological and immunohistochemical studies of the proximal caecum and caecal tonsils of quail (Coturnix coturnix japonica).Anat Histol Embryol. 2019 Sep;48(5):476-485. doi: 10.1111/ahe.12469. Epub 2019 Jul 15. Anat Histol Embryol. 2019. PMID: 31305954
-
Structure of the oesophagus and morphometric, histochemical-immunohistochemical profiles of the oesophageal gland during the post-hatching period of Japanese quails (Coturnix coturnix japonica).Anat Histol Embryol. 2009 Oct;38(5):330-40. doi: 10.1111/j.1439-0264.2009.00947.x. Epub 2009 Jul 27. Anat Histol Embryol. 2009. PMID: 19650777
-
Developmental events in the lung of the Japanese quail (Coturnix coturnix japonica): Morphological, histochemical and electron-microscopic studies.Microsc Res Tech. 2022 Dec;85(12):3761-3776. doi: 10.1002/jemt.24225. Epub 2022 Aug 24. Microsc Res Tech. 2022. PMID: 36053915
Cited by
-
Influence of Heat Treatment on Muscle Recovery after Skeletal Muscle Injury in Rats: Histological and Immunohistochemical Studies.J Microsc Ultrastruct. 2021 Jun 3;10(2):63-71. doi: 10.4103/jmau.jmau_85_20. eCollection 2022 Apr-Jun. J Microsc Ultrastruct. 2021. PMID: 35832314 Free PMC article.
-
Teleost innate immunity, an intricate game between immune cells and parasites of fish organs: who wins, who loses.Front Immunol. 2023 Oct 16;14:1250835. doi: 10.3389/fimmu.2023.1250835. eCollection 2023. Front Immunol. 2023. PMID: 37908358 Free PMC article. Review.
-
Revealing the physical restrictions of caecal influx in broilers through the use of solid and soluble markers.Anim Nutr. 2025 Jan 16;21:37-48. doi: 10.1016/j.aninu.2024.12.004. eCollection 2025 Jun. Anim Nutr. 2025. PMID: 40135172 Free PMC article.
-
Descriptive embryological insights of the colorectum of quail embryos with concern to its functional morphology.BMC Vet Res. 2024 Nov 6;20(1):508. doi: 10.1186/s12917-024-04341-z. BMC Vet Res. 2024. PMID: 39506803 Free PMC article.
-
Developmental characteristics of cutaneous telocytes in late embryos of the silky fowl.Eur J Histochem. 2024 Oct 15;68(4):4089. doi: 10.4081/ejh.2024.4089. Eur J Histochem. 2024. PMID: 39410819 Free PMC article.
References
-
- Zacchei AM. The embryonal development of the Japanese quail (Coturnix coturnix japonica) Arch. Ital. Anat. Embryol. 1961;66:36–62. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

