Peripheral nerve magnetic resonance imaging
- PMID: 31700612
- PMCID: PMC6820826
- DOI: 10.12688/f1000research.19695.1
Peripheral nerve magnetic resonance imaging
Abstract
Magnetic resonance imaging (MRI) has been used extensively in revealing pathological changes in the central nervous system. However, to date, MRI is very much underutilized in evaluating the peripheral nervous system (PNS). This underutilization is generally due to two perceived weaknesses in MRI: first, the need for very high resolution to image the small structures within the peripheral nerves to visualize morphological changes; second, the lack of normative data in MRI of the PNS and this makes reliable interpretation of the data difficult. This article reviews current state-of-the-art capabilities in in vivo MRI of human peripheral nerves. It aims to identify areas where progress has been made and those that still require further improvement. In particular, with many new therapies on the horizon, this review addresses how MRI can be used to provide non-invasive and objective biomarkers in the evaluation of peripheral neuropathies. Although a number of techniques are available in diagnosing and tracking pathologies in the PNS, those techniques typically target the distal peripheral nerves, and distal nerves may be completely degenerated during the patient's first clinic visit. These techniques may also not be able to access the proximal nerves deeply embedded in the tissue. Peripheral nerve MRI would be an alternative to circumvent these problems. In order to address the pressing clinical needs, this review closes with a clinical protocol at 3T that will allow high-resolution, high-contrast, quantitative MRI of the proximal peripheral nerves.
Keywords: Charcot-Marie-Tooth Disease; Magnetic Resonance Imaging; Peripheral Nerves; Peripheral Nervous System; Peripheral Neuropathy; Sciatic Nerve.
Copyright: © 2019 Chen Y et al.
Conflict of interest statement
No competing interests were disclosed.No competing interests were disclosed.No competing interests were disclosed.
Figures
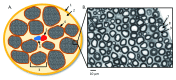


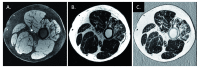
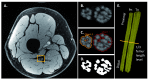
References
-
- Marieb EN, Hoehn K: Human anatomy & physiology. Pearson Education;2007. Reference Source
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

