SKA-31, an activator of Ca2+-activated K+ channels, improves cardiovascular function in aging
- PMID: 31707036
- PMCID: PMC6956598
- DOI: 10.1016/j.phrs.2019.104539
SKA-31, an activator of Ca2+-activated K+ channels, improves cardiovascular function in aging
Abstract
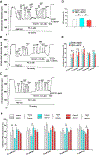
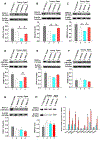
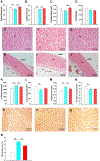
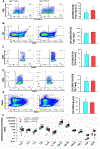
Aging represents an independent risk factor for the development of cardiovascular disease, and is associated with complex structural and functional alterations in the vasculature, such as endothelial dysfunction. Small- and intermediate-conductance, Ca2+-activated K+ channels (KCa2.3 and KCa3.1, respectively) are prominently expressed in the vascular endothelium, and pharmacological activators of these channels induce robust vasodilation upon acute exposure in isolated arteries and intact animals. However, the effects of prolonged in vivo administration of such compounds are unknown. In our study, we hypothesized that such treatment would ameliorate aging-related cardiovascular deficits. Aged (∼18 months) male Sprague Dawley rats were treated daily with either vehicle or the KCa channel activator SKA-31 (10 mg/kg, intraperitoneal injection; n = 6/group) for 8 weeks, followed by echocardiography, arterial pressure myography, immune cell and plasma cytokine characterization, and tissue histology. Our results show that SKA-31 administration improved endothelium-dependent vasodilation, reduced agonist-induced vascular contractility, and prevented the aging-associated declines in cardiac ejection fraction, stroke volume and fractional shortening, and further improved the expression of endothelial KCa channels and associated cell signalling components to levels similar to those observed in young male rats (∼5 months at end of study). SKA-31 administration did not promote pro-inflammatory changes in either T cell populations or plasma cytokines/chemokines, and we observed no overt tissue histopathology in heart, kidney, aorta, brain, liver and spleen. SKA-31 treatment in young rats had little to no effect on vascular reactivity, select protein expression, tissue histology, plasma cytokines/chemokines or immune cell properties. Collectively, these data demonstrate that administration of the KCa channel activator SKA-31 improved aging-related cardiovascular function, without adversely affecting the immune system or promoting tissue toxicity.
Keywords: Aging; Arteries; Ca(2+)-activated K(+) channels; Heart; Immune system.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Disclosures
The authors declare that they have no disclosures or competing financial interests related to this study.
Figures

Similar articles
-
SKA-31, an activator of endothelial Ca2+-activated K+ channels evokes robust vasodilation in rat mesenteric arteries.Eur J Pharmacol. 2018 Jul 15;831:60-67. doi: 10.1016/j.ejphar.2018.05.006. Epub 2018 May 9. Eur J Pharmacol. 2018. PMID: 29753043
-
Modulation of Cardiovascular Function in Primary Hypertension in Rat by SKA-31, an Activator of KCa2.x and KCa3.1 Channels.Int J Mol Sci. 2019 Aug 23;20(17):4118. doi: 10.3390/ijms20174118. Int J Mol Sci. 2019. PMID: 31450834 Free PMC article.
-
Inhibition of Myogenic Tone in Rat Cremaster and Cerebral Arteries by SKA-31, an Activator of Endothelial KCa2.3 and KCa3.1 Channels.J Cardiovasc Pharmacol. 2015 Jul;66(1):118-27. doi: 10.1097/FJC.0000000000000252. J Cardiovasc Pharmacol. 2015. PMID: 25815673 Free PMC article.
-
Pharmacologic targeting of endothelial Ca2+-activated K+ channels: A strategy to improve cardiovascular function.Channels (Austin). 2018 Jan 1;12(1):126-136. doi: 10.1080/19336950.2018.1454814. Channels (Austin). 2018. PMID: 29577810 Free PMC article. Review.
-
Vascular KCa-channels as therapeutic targets in hypertension and restenosis disease.Expert Opin Ther Targets. 2010 Feb;14(2):143-55. doi: 10.1517/14728220903540257. Expert Opin Ther Targets. 2010. PMID: 20055714 Free PMC article. Review.
Cited by
-
Development of Alzheimer's Disease Progressively Alters Sex-Dependent KCa and Sex-Independent KIR Channel Function in Cerebrovascular Endothelium.J Alzheimers Dis. 2020;76(4):1423-1442. doi: 10.3233/JAD-200085. J Alzheimers Dis. 2020. PMID: 32651315 Free PMC article.
-
Research progress on the mechanism of aging of vascular endothelial cells and the intervention of traditional Chinese medicine: A review.Medicine (Baltimore). 2022 Dec 9;101(49):e32248. doi: 10.1097/MD.0000000000032248. Medicine (Baltimore). 2022. PMID: 36626478 Free PMC article. Review.
-
The novel quinoline derivative SKA-346 as a KCa3.1 channel selective activator.RSC Adv. 2024 Dec 4;14(52):38364-38377. doi: 10.1039/d4ra07330d. eCollection 2024 Dec 3. RSC Adv. 2024. PMID: 39635364 Free PMC article.
-
Regulatory role of KCa3.1 in immune cell function and its emerging association with rheumatoid arthritis.Front Immunol. 2022 Oct 5;13:997621. doi: 10.3389/fimmu.2022.997621. eCollection 2022. Front Immunol. 2022. PMID: 36275686 Free PMC article. Review.
-
KCa 2.2 (KCNN2): A physiologically and therapeutically important potassium channel.J Neurosci Res. 2023 Nov;101(11):1699-1710. doi: 10.1002/jnr.25233. Epub 2023 Jul 19. J Neurosci Res. 2023. PMID: 37466411 Free PMC article. Review.
References
-
- Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation. 2003;107(1):139–146. - PubMed
-
- Dai Z, Aoki T, Fukumoto Y, Shimokawa H. Coronary perivascular fibrosis is associated with impairment of coronary blood flow in patients with non-ischemic heart failure. J. Cardiol 2012;60(5):416–421. - PubMed
-
- Gates PE, Strain WD, Shore AC. Human endothelial function and microvascular ageing. Experimental Physiology 2009;94(3):311–316. - PubMed

