Computational Analysis of Therapeutic Neuroadaptation to Chronic Antidepressant in a Model of the Monoaminergic Neurotransmitter and Stress Hormone Systems
- PMID: 31708770
- PMCID: PMC6823241
- DOI: 10.3389/fphar.2019.01215
Computational Analysis of Therapeutic Neuroadaptation to Chronic Antidepressant in a Model of the Monoaminergic Neurotransmitter and Stress Hormone Systems
Abstract
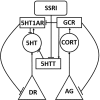
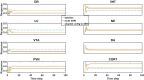
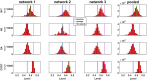
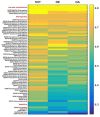
The clinical practice of selective serotonin reuptake inhibitor (SSRI) augmentation relies heavily on trial-and-error. Unfortunately, the drug combinations prescribed today fail to provide relief for many depressed patients. In order to identify potentially more effective treatments, we developed a computational model of the monoaminergic neurotransmitter and stress-steroid systems that neuroadapts to chronic administration of combinations of antidepressant drugs and hormones by adjusting the strengths of its transmitter-system components (TSCs). We used the model to screen 60 chronically administered drug/hormone pairs and triples, and identified as potentially therapeutic those combinations that raised the monoamines (serotonin, norepinephrine, and dopamine) but lowered cortisol following neuroadaptation in the model. We also evaluated the contributions of individual and pairs of TSCs to therapeutic neuroadaptation with chronic SSRI using sensitivity, correlation, and linear temporal-logic analyses. All three approaches revealed that therapeutic neuroadaptation to chronic SSRI is an overdetermined process that depends on multiple TSCs, providing a potential explanation for the clinical finding that no single antidepressant regimen alleviates depressive symptoms in all patients.
Keywords: SSRI augmentation; combination therapy; cortisol; depression; monoamine; overdetermined system; polypharmacy; systems biology.
Copyright © 2019 Camacho, Vijitbenjaronk and Anastasio.
Figures



Similar articles
-
Computational modeling of the monoaminergic neurotransmitter and male neuroendocrine systems in an analysis of therapeutic neuroadaptation to chronic antidepressant.Eur Neuropsychopharmacol. 2020 Feb;31:86-99. doi: 10.1016/j.euroneuro.2019.11.003. Epub 2019 Dec 9. Eur Neuropsychopharmacol. 2020. PMID: 31831204
-
Computational Model of Antidepressant Response Heterogeneity as Multi-pathway Neuroadaptation.Front Pharmacol. 2017 Dec 20;8:925. doi: 10.3389/fphar.2017.00925. eCollection 2017. Front Pharmacol. 2017. PMID: 29375372 Free PMC article.
-
Pharmacologic approaches to treatment resistant depression: Evidences and personal experience.World J Psychiatry. 2015 Sep 22;5(3):330-41. doi: 10.5498/wjp.v5.i3.330. World J Psychiatry. 2015. PMID: 26425446 Free PMC article.
-
Monoamine neurocircuitry in depression and strategies for new treatments.Prog Neuropsychopharmacol Biol Psychiatry. 2013 Aug 1;45:54-63. doi: 10.1016/j.pnpbp.2013.04.009. Epub 2013 Apr 19. Prog Neuropsychopharmacol Biol Psychiatry. 2013. PMID: 23602950 Review.
-
[Mechanism of action of antidepressants and therapeutic perspectives].Therapie. 2002 Jul-Aug;57(4):385-96. Therapie. 2002. PMID: 12422559 Review. French.
References
-
- Amini-Khoei H., Mohammadi-Asl A., Amiri S., Hosseini M.-J., Momeny M., Hassanipour M., et al. (2017). Oxytocin mitigated the depressive-like behaviors of maternal separation stress through modulating mitochondrial function and neuroinflammation. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 76, 169–178. 10.1016/j.pnpbp.2017.02.022 - DOI - PubMed
LinkOut - more resources
Full Text Sources

