Eriocalyxin B Induces Apoptosis and Autophagy Involving Akt/Mammalian Target of Rapamycin (mTOR) Pathway in Prostate Cancer Cells
- PMID: 31714902
- PMCID: PMC6873644
- DOI: 10.12659/MSM.917333
Eriocalyxin B Induces Apoptosis and Autophagy Involving Akt/Mammalian Target of Rapamycin (mTOR) Pathway in Prostate Cancer Cells
Abstract
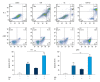
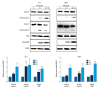
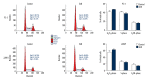
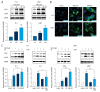
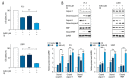
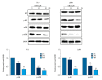
BACKGROUND Eriocalyxin B (EriB), a diterpenoid isolated from the plant Isodon eriocalyx, has been shown to possess anti-tumor properties. However, few systematic studies of the mechanism underlying the anti-tumor activity of Eriocalyxin B in prostate cancer cells have been published. The aim of this study was to investigate the effect of Eriocalyxin B on prostate cancer cells. MATERIAL AND METHODS In the present study, the PC-3 (androgen-independent) and 22RV1 (androgen-dependent) human prostate cancer cell lines were cultured with and without increasing doses of Eriocalyxin B. MTT assay was used to measure cell viability. Western blotting was performed to measure levels of proteins associated with apoptosis and autophagy. Flow cytometry was used to assess changes in cell apoptosis and cycle. Fluorescence microscopy was used to capture images of autophagy-related proteins. RESULTS Treatment of human prostate cancer cells with Eriocalyxin B resulted in apoptosis in a dose- and time-dependent manner. Eriocalyxin B also induced autophagy, with elevated LC3B-II protein expression and punctuate patterns. Additionally, autophagy protected prostate cancer cells from apoptosis induced by Eriocalyxin B, which was demonstrated by addition of chloroquine (CQ). Moreover, the results indicated that Eriocalyxin B could inhibit the phosphorylation of Akt and mTOR. Eriocalyxin B induced apoptosis and autophagy by inhibition of the Akt/mTOR pathway. CONCLUSIONS Eriocalyxin B induces apoptosis and autophagy involving the Akt/mTOR pathway in prostate cancer cells in vitro. These findings provide evidence for Eriocalyxin B as a potent therapeutic for the treatment of prostate cancer.
Conflict of interest statement
None.
Figures
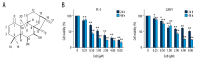
References
-
- Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. part 1: Screening, diagnosis, and local treatment with curative intent. Eur Urol. 2016;71:618–29. - PubMed
-
- Alva A, Hussain M. Intermittent androgen deprivation therapy in advanced prostate cancer. Curr Treat Options Oncol. 2014;15:127–36. - PubMed
-
- Tanaka S, Hikita H, Tatsumi T, et al. Rubicon inhibits autophagy and accelerates hepatocyte apoptosis and lipid accumulation in nonalcoholic fatty liver disease in mice. Hepatology. 2016;64:1994–2014. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

