N-Dihydrogalactochitosan Potentiates the Radiosensitivity of Liver Metastatic Tumor Cells Originated from Murine Breast Tumors
- PMID: 31717306
- PMCID: PMC6888949
- DOI: 10.3390/ijms20225581
N-Dihydrogalactochitosan Potentiates the Radiosensitivity of Liver Metastatic Tumor Cells Originated from Murine Breast Tumors
Abstract
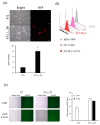
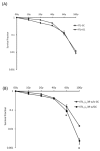
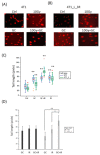
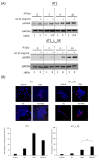
Radiation is a widely used therapeutic method for treating breast cancer. N-dihydrogalactochitosan (GC), a biocompatible immunostimulant, is known to enhance the effects of various treatment modalities in different tumor types. However, whether GC can enhance the radiosensitivity of cancer cells remains to be explored. In this study, triple-negative murine 4T1 breast cancer cells transduced with multi-reporter genes were implanted in immunocompetent Balb/C mice to track, dissect, and identify liver-metastatic 4T1 cells. These cells expressed cancer stem cell (CSC) -related characteristics, including the ability to form spheroids, the expression of the CD44 marker, and the increase of protein stability. We then ex vivo investigated the potential effect of GC on the radiosensitivity of the liver-metastatic 4T1 breast cancer cells and compared the results to those of parental 4T1 cells subjected to the same treatment. The cells were irradiated with increased doses of X-rays with or without GC treatment. Colony formation assays were then performed to determine the survival fractions and radiosensitivity of these cells. We found that GC preferably increased the radiosensitivity of liver-metastatic 4T1 breast cancer cells rather than that of the parental cells. Additionally, the single-cell DNA electrophoresis assay (SCDEA) and γ-H2AX foci assay were performed to assess the level of double-stranded DNA breaks (DSBs). Compared to the parental cells, DNA damage was significantly increased in liver-metastatic 4T1 cells after they were treated with GC plus radiation. Further studies on apoptosis showed that this combination treatment increased the sub-G1 population of cells, but not caspase-3 cleavage, in liver-metastatic breast cancer cells. Taken together, the current data suggest that the synergistic effects of GC and irradiation might be used to enhance the efficacy of radiotherapy in treating metastatic tumors.
Keywords: N-dihydrogalactochitosan (GC), metastatic tumors; cancer stem cells; radiosensitivity; triple-negative breast cancer.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

Similar articles
-
Targeting BRG1 chromatin remodeler via its bromodomain for enhanced tumor cell radiosensitivity in vitro and in vivo.Mol Cancer Ther. 2015 Feb;14(2):597-607. doi: 10.1158/1535-7163.MCT-14-0372. Epub 2014 Dec 12. Mol Cancer Ther. 2015. PMID: 25504753
-
Sonic Hedgehog inhibition as a strategy to augment radiosensitivity of hepatocellular carcinoma.J Gastroenterol Hepatol. 2015 Aug;30(8):1317-24. doi: 10.1111/jgh.12931. J Gastroenterol Hepatol. 2015. PMID: 25682950
-
Targeting PI3K and AMPKα Signaling Alone or in Combination to Enhance Radiosensitivity of Triple Negative Breast Cancer.Cells. 2020 May 19;9(5):1253. doi: 10.3390/cells9051253. Cells. 2020. PMID: 32438621 Free PMC article.
-
Use of the γ-H2AX assay to monitor DNA damage and repair in translational cancer research.Cancer Lett. 2012 Dec 31;327(1-2):123-33. doi: 10.1016/j.canlet.2011.12.025. Epub 2011 Dec 21. Cancer Lett. 2012. PMID: 22198208 Free PMC article. Review.
-
Reporting of methodologies used for clonogenic assays to determine radiosensitivity.J Radiat Res. 2020 Nov 16;61(6):828-831. doi: 10.1093/jrr/rraa064. J Radiat Res. 2020. PMID: 32823284 Free PMC article. Review.
Cited by
-
The pathogenesis of liver cancer and the therapeutic potential of bioactive substances.Front Pharmacol. 2022 Oct 5;13:1029601. doi: 10.3389/fphar.2022.1029601. eCollection 2022. Front Pharmacol. 2022. PMID: 36278230 Free PMC article. Review.
-
Enhancing the Immunotherapeutic Effect by IP-001 and Irreversible Electroporation in Mouse Oligometastatic Models of Pancreatic Adenocarcinoma.Ann Surg Oncol. 2025 Apr;32(4):2786-2798. doi: 10.1245/s10434-024-16742-3. Epub 2024 Dec 29. Ann Surg Oncol. 2025. PMID: 39739260
-
Editorial to Radiation in Multimodal Tumor Immune Therapies-Mechanisms and Application.Int J Mol Sci. 2021 Jul 17;22(14):7648. doi: 10.3390/ijms22147648. Int J Mol Sci. 2021. PMID: 34299268 Free PMC article.
-
BPR0C261, An Analogous of Microtubule Disrupting Agent D-24851 Enhances the Radiosensitivity of Human Non-Small Cell Lung Cancer Cells via p53-Dependent and p53-Independent Pathways.Int J Mol Sci. 2022 Nov 15;23(22):14083. doi: 10.3390/ijms232214083. Int J Mol Sci. 2022. PMID: 36430560 Free PMC article.
-
Radiosensitizing Effect of Celastrol by Inhibiting G2/M Phase Arrest Induced by the c-myc Gene of Human SW1353 Chondrosarcoma Cells: Network and Experimental Analyses.Biomed Res Int. 2022 Jan 31;2022:1948657. doi: 10.1155/2022/1948657. eCollection 2022. Biomed Res Int. 2022. Retraction in: Biomed Res Int. 2024 Mar 20;2024:9784090. doi: 10.1155/2024/9784090. PMID: 35141331 Free PMC article. Retracted.
References
-
- Haug A.R., Tiega Donfack B.P., Trumm C., Zech C.J., Michl M., Laubender R.P., Uebleis C., Bartenstein P., Heinemann V., Hacker M. 18F-FDG PET/CT predicts survival after radioembolization of hepatic metastases from breast cancer. J. Nucl. Med. 2012;53:371–377. doi: 10.2967/jnumed.111.096230. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

