Nav1.6 promotes inflammation and neuronal degeneration in a mouse model of multiple sclerosis
- PMID: 31722722
- PMCID: PMC6852902
- DOI: 10.1186/s12974-019-1622-1
Nav1.6 promotes inflammation and neuronal degeneration in a mouse model of multiple sclerosis
Abstract
Background: In multiple sclerosis (MS) and in the experimental autoimmune encephalomyelitis (EAE) model of MS, the Nav1.6 voltage-gated sodium (Nav) channel isoform has been implicated as a primary contributor to axonal degeneration. Following demyelination Nav1.6, which is normally co-localized with the Na+/Ca2+ exchanger (NCX) at the nodes of Ranvier, associates with β-APP, a marker of neural injury. The persistent influx of sodium through Nav1.6 is believed to reverse the function of NCX, resulting in an increased influx of damaging Ca2+ ions. However, direct evidence for the role of Nav1.6 in axonal degeneration is lacking.
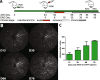
Methods: In mice floxed for Scn8a, the gene that encodes the α subunit of Nav1.6, subjected to EAE we examined the effect of eliminating Nav1.6 from retinal ganglion cells (RGC) in one eye using an AAV vector harboring Cre and GFP, while using the contralateral either injected with AAV vector harboring GFP alone or non-targeted eye as control.
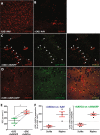
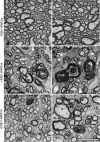
Results: In retinas, the expression of Rbpms, a marker for retinal ganglion cells, was found to be inversely correlated to the expression of Scn8a. Furthermore, the gene expression of the pro-inflammatory cytokines Il6 (IL-6) and Ifng (IFN-γ), and of the reactive gliosis marker Gfap (GFAP) were found to be reduced in targeted retinas. Optic nerves from targeted eyes were shown to have reduced macrophage infiltration and improved axonal health.
Conclusion: Taken together, our results are consistent with Nav1.6 promoting inflammation and contributing to axonal degeneration following demyelination.
Keywords: Adeno-associated virus; Conditional knockout; Experimental autoimmune encephalitis; Inflammation; Multiple sclerosis; Nav1.6; Optic nerve; Optic neuritis; Retinal ganglion cells; Scn8a; Sodium channel.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



References
-
- Mohr DC. Psychiatric Disorders, Stress, and Their Treatment Among People with Multiple Sclerosis. Psychol Co-morbidities Phys Illn. 2011:311–34.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

