Maintenance Therapy With Interleukin-2 for Childhood AML: Results of ELAM02 Phase III Randomized Trial
- PMID: 31723797
- PMCID: PMC6745961
- DOI: 10.1097/HS9.0000000000000159
Maintenance Therapy With Interleukin-2 for Childhood AML: Results of ELAM02 Phase III Randomized Trial
Abstract
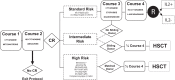
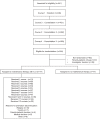
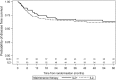
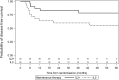
Despite significant progress in the treatment of pediatric acute myeloblastic leukemia (AML), relapse remains the commonest cause of death. Randomized ELAM02 trial questioned if maintenance therapy with interleukin-2 (IL2), for 1 year, improves disease-free survival (DFS). Patients aged 0 to 18 years, with newly diagnosed AML (excluding patients with acute promyelocytic leukemia or down syndrome AML) were enrolled. They received 1 course of induction treatment (cytarabine and mitoxantrone) and 3 courses of consolidation treatment (high-dose cytarabine in courses 1 and 3). According to the cytogenetics risk, patients not undergoing hematopoietic stem cell transplantation, still in complete remission (CR) after the third course of consolidation treatment, were eligible for randomization to 1 year of maintenance therapy with monthly courses of IL2 or no maintenance treatment. There were 438 evaluable patients, 154 of whom were randomized to the IL2/no maintenance groups. Relapse occurred in 28 patients from the IL2+ group and 29 patients in the IL2- group. Survival was similar in the 2 groups, with a 4-year DFS of 62% without IL2 and 66% with IL2 (P = 0.75). In the CBF population, 4-year DFS was 55% without IL2 and 78% with IL2 (P = 0.07). No deaths from toxicity or excess of serious adverse events related to IL2 treatment were recorded. Prolonged IL2 for maintenance therapy after intensive chemotherapy is feasible and safe in pediatric AML patients in their first CR. Such treatment did not improve DFS in this study, but a positive trend was observed in favor of IL2 maintenance therapy among core binding factor acute myeloblastic leukemia.
Copyright © 2018 the Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the European Hematology Association.
Figures

References
-
- Clavel J, Goubin A, Auclerc MF, et al. Incidence of childhood leukaemia and non-Hodgkin's lymphoma in France: National Registry of Childhood Leukaemia and Lymphoma, 1990–1999. Eur J Cancer Prev. 2004;13:97–103. - PubMed
-
- Gibson BE, Webb DK, Howman AJ, et al. Results of a randomized trial in children with acute myeloid leukaemia: medical research council AML12 trial. Br J Haematol. 2011;155:366–376. - PubMed
-
- Creutzig U, Zimmermann M, Bourquin JP, et al. Randomized trial comparing liposomal daunorubicin with Idarubicin as induction for pediatric acute myeloid leukemia: results from study AML-BFM 2004. Blood. 2013;122:37–43. - PubMed
-
- Gamis AS, Alonzo TA, Meshinchi S, et al. Gemtuzumab ozogamicin in children and adolescents with de novo acute myeloid leukemia improves event-free survival by reducing relapse risk: results from the randomized phase III Children's Oncology Group trial AAML0531. J Clin Oncol. 2014;32:3021–3032. - PMC - PubMed
-
- Perel Y, Auvrignon A, Leblanc T, et al. Treatment of childhood acute myeloblastic leukemia: dose intensification improves outcome and maintenance therapy is of no benefit—multicenter studies of the French LAME (Leucemie Aigue Myeloblastique Enfant) Cooperative Group. Leukemia. 2005;19:2082–2089. - PubMed
LinkOut - more resources
Full Text Sources
