Pathways to defense metabolites and evading fruit bitterness in genus Solanum evolved through 2-oxoglutarate-dependent dioxygenases
- PMID: 31727889
- PMCID: PMC6856131
- DOI: 10.1038/s41467-019-13211-4
Pathways to defense metabolites and evading fruit bitterness in genus Solanum evolved through 2-oxoglutarate-dependent dioxygenases
Abstract
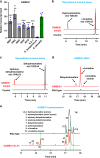
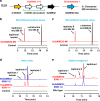
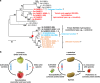
The genus Solanum comprises three food crops (potato, tomato, and eggplant), which are consumed on daily basis worldwide and also producers of notorious anti-nutritional steroidal glycoalkaloids (SGAs). Hydroxylated SGAs (i.e. leptinines) serve as precursors for leptines that act as defenses against Colorado Potato Beetle (Leptinotarsa decemlineata Say), an important pest of potato worldwide. However, SGA hydroxylating enzymes remain unknown. Here, we discover that 2-OXOGLUTARATE-DEPENDENT-DIOXYGENASE (2-ODD) enzymes catalyze SGA-hydroxylation across various Solanum species. In contrast to cultivated potato, Solanum chacoense, a widespread wild potato species, has evolved a 2-ODD enzyme leading to the formation of leptinines. Furthermore, we find a related 2-ODD in tomato that catalyzes the hydroxylation of the bitter α-tomatine to hydroxytomatine, the first committed step in the chemical shift towards downstream ripening-associated non-bitter SGAs (e.g. esculeoside A). This 2-ODD enzyme prevents bitterness in ripe tomato fruit consumed today which otherwise would remain unpleasant in taste and more toxic.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Friedman M, McDonald G, Filadelfi-keszi M. Potato glycoalkaloids: chemistry analysis safety and plant physiology. Crit. Rev. Plant Sci. 1997;16:55–132. doi: 10.1080/07352689709701946. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

