Polymer Membranes Sonocoated and Electrosprayed with Nano-Hydroxyapatite for Periodontal Tissues Regeneration
- PMID: 31731775
- PMCID: PMC6915502
- DOI: 10.3390/nano9111625
Polymer Membranes Sonocoated and Electrosprayed with Nano-Hydroxyapatite for Periodontal Tissues Regeneration
Abstract
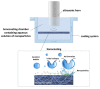
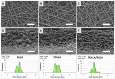
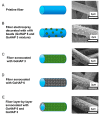
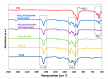
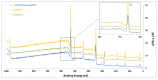
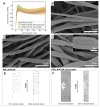
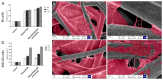
Diseases of periodontal tissues are a considerable clinical problem, connected with inflammatory processes and bone loss. The healing process often requires reconstruction of lost bone in the periodontal area. For that purpose, various membranes are used to prevent ingrowth of epithelium in the tissue defect and enhance bone regeneration. Currently-used membranes are mainly non-resorbable or are derived from animal tissues. Thus, there is an urgent need for non-animal-derived bioresorbable membranes with tuned resorption rates and porosity optimized for the circulation of body nutrients. We demonstrate membranes produced by the electrospinning of biodegradable polymers (PDLLA/PLGA) coated with nanohydroxyapatite (nHA). The nHA coating was made using two methods: sonocoating and electrospraying of nHA suspensions. In a simulated degradation study, for electrosprayed membranes, short-term calcium release was observed, followed by hydrolytic degradation. Sonocoating produced a well-adhering nHA layer with full coverage of the fibers. The layer slowed the polymer degradation and increased the membrane wettability. Due to gradual release of calcium ions the degradation-associated acidity of the polymer was neutralized. The sonocoated membranes exhibited good cellular metabolic activity responses against MG-63 and BJ cells. The collected results suggest their potential use in Guided Tissue Regeneration (GTR) and Guided Bone Regeneration (GBR) periodontal procedures.
Keywords: GTR/GBR membranes; electrospinning; electrospraying; periodontal regeneration; sonocoating.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Electrospun Membrane Surface Modification by Sonocoating with HA and ZnO:Ag Nanoparticles-Characterization and Evaluation of Osteoblasts and Bacterial Cell Behavior In Vitro.Cells. 2022 May 8;11(9):1582. doi: 10.3390/cells11091582. Cells. 2022. PMID: 35563888 Free PMC article.
-
Freeze gelated porous membranes for periodontal tissue regeneration.Acta Biomater. 2015 Sep;23:317-328. doi: 10.1016/j.actbio.2015.05.001. Epub 2015 May 9. Acta Biomater. 2015. PMID: 25968357
-
A biodegradable multifunctional nanofibrous membrane for periodontal tissue regeneration.Acta Biomater. 2020 May;108:207-222. doi: 10.1016/j.actbio.2020.03.044. Epub 2020 Apr 3. Acta Biomater. 2020. PMID: 32251784
-
Collagen based barrier membranes for periodontal guided bone regeneration applications.Odontology. 2017 Jan;105(1):1-12. doi: 10.1007/s10266-016-0267-0. Epub 2016 Sep 9. Odontology. 2017. PMID: 27613193 Review.
-
Asymmetric resorbable-based dental barrier membrane for periodontal guided tissue regeneration and guided bone regeneration: A review.J Biomed Mater Res B Appl Biomater. 2022 Sep;110(9):2157-2182. doi: 10.1002/jbm.b.35060. Epub 2022 Mar 24. J Biomed Mater Res B Appl Biomater. 2022. PMID: 35322931 Review.
Cited by
-
Drug-Releasing Antibacterial Coating Made from Nano-Hydroxyapatite Using the Sonocoating Method.Nanomaterials (Basel). 2021 Jun 28;11(7):1690. doi: 10.3390/nano11071690. Nanomaterials (Basel). 2021. PMID: 34203218 Free PMC article.
-
Advances of Oxidative Stress Impact in Periodontitis: Biomarkers and Effective Targeting Options.Curr Med Chem. 2024;31(38):6187-6203. doi: 10.2174/0109298673297545240507091410. Curr Med Chem. 2024. PMID: 38726786 Review.
-
Applications and interventions of polymers and nanomaterials in alveolar bone regeneration and tooth dentistry.RSC Adv. 2024 Nov 12;14(49):36226-36245. doi: 10.1039/d4ra06092j. eCollection 2024 Nov 11. RSC Adv. 2024. PMID: 39534053 Free PMC article. Review.
-
Electrospun Membrane Surface Modification by Sonocoating with HA and ZnO:Ag Nanoparticles-Characterization and Evaluation of Osteoblasts and Bacterial Cell Behavior In Vitro.Cells. 2022 May 8;11(9):1582. doi: 10.3390/cells11091582. Cells. 2022. PMID: 35563888 Free PMC article.
-
Effective Targeting of Colon Cancer Cells with Piperine Natural Anticancer Prodrug Using Functionalized Clusters of Hydroxyapatite Nanoparticles.Pharmaceutics. 2020 Jan 16;12(1):70. doi: 10.3390/pharmaceutics12010070. Pharmaceutics. 2020. PMID: 31963155 Free PMC article.
References
-
- Cierech M., Kolenda A., Grudniak A.M., Wojnarowicz J., Woźniak B., Gołaś M., Swoboda-Kopeć E., Łojkowski W., Mierzwińska-Nastalska E. Significance of polymethylmethacrylate (PMMA) modification by zinc oxide nanoparticles for fungal biofilm formation. Int. J. Pharm. 2016;510:323–335. doi: 10.1016/j.ijpharm.2016.06.052. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

