Use of hPSC-derived 3D organoids and mouse genetics to define the roles of YAP in the development of the esophagus
- PMID: 31748205
- PMCID: PMC6918786
- DOI: 10.1242/dev.178855
Use of hPSC-derived 3D organoids and mouse genetics to define the roles of YAP in the development of the esophagus
Abstract
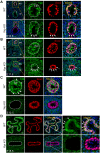
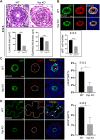
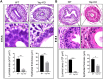
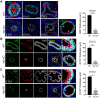
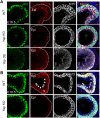
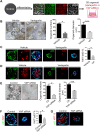
Balanced progenitor activities are crucial for the development and maintenance of high turn-over organs such as the esophagus. However, the molecular mechanisms regulating these progenitor activities in the esophagus remain to be elucidated. Here, we demonstrated that Yap is required for the proliferation of esophageal progenitor cells (EPCs) in the developing murine esophagus. We found that Yap deficiency reduces EPC proliferation and stratification whereas persistent Yap activation increases cell proliferation and causes aberrant stratification of the developing esophagus. We further demonstrated that the role of YAP signaling is conserved in the developing human esophagus by utilizing 3D human pluripotent stem cell (hPSC)-derived esophageal organoid culture. Taken together, our studies combining loss/gain-of-function murine models and hPSC differentiation support a key role for YAP in the self-renewal of EPCs and stratification of the esophageal epithelium.
Keywords: 3D organoids; Esophagus; Hippo pathway; Human pluripotent stem cells; Progenitor cells; YAP.
© 2019. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interestsThe authors declare no competing or financial interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

