Proteomics-based screening of the target proteins associated with antidepressant-like effect and mechanism of Saikosaponin A
- PMID: 31762213
- PMCID: PMC6933357
- DOI: 10.1111/jcmm.14695
Proteomics-based screening of the target proteins associated with antidepressant-like effect and mechanism of Saikosaponin A
Abstract
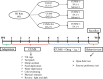
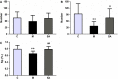
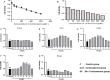
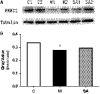
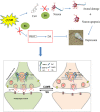
Depression is a commonly occurring neuropsychiatric disease with an increasing incidence rate. Saikosaponin A (SA), a major bioactive component extracted from Radix Bupleuri, possesses anti-malignant cell proliferation, anti-inflammation, anti-oxidation and liver protective effects. However, few studies have investigated SA's antidepressant effects and pharmacological mechanisms of action. Our study aimed to explore the anti-depression effect of SA and screen the target proteins regulated by SA in a rat model of chronic unpredictable mild stress (CUMS)-induced depression. Results showed that 8-week CUMS combined with separation could successfully produce depressive-like behaviours and cause a decrease of dopamine (DA) in rat hippocampus, and 4-week administration of SA could relieve CUMS rats' depressive symptoms and up-regulated DA content. There were 15 kinds of significant differentially expressed proteins that were detected not only between the control and CUMS groups, but also between the CUMS and SA treatment groups. Proline-rich transmembrane protein 2 (PRRT2) was down-regulated by CUMS while up-regulated by SA. These findings reveal that SA may exert antidepressant effects by up-regulating the expression level of PRRT2 and increasing DA content in hippocampus. The identification of these 15 differentially expressed proteins, including PRRT2, provides further insight into the treatment mechanism of SA for depression.
Keywords: Saikosaponin A; depression; dopamine; proline-rich transmembrane protein 2; proteomics.
© 2019 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Conflict of interest statement
The authors confirm that there are no conflicts of interest.
Figures




Similar articles
-
Saikosaponin A attenuates perimenopausal depression-like symptoms by chronic unpredictable mild stress.Neurosci Lett. 2018 Jan 1;662:283-289. doi: 10.1016/j.neulet.2017.09.046. Epub 2017 Sep 25. Neurosci Lett. 2018. PMID: 28958685
-
Saikosaponin A improved depression-like behavior and inhibited hippocampal neuronal apoptosis after cerebral ischemia through p-CREB/BDNF pathway.Behav Brain Res. 2021 Apr 9;403:113138. doi: 10.1016/j.bbr.2021.113138. Epub 2021 Jan 22. Behav Brain Res. 2021. PMID: 33493495
-
Antidepressant-like effects of Xiaochaihutang in a rat model of chronic unpredictable mild stress.J Ethnopharmacol. 2014 Feb 27;152(1):217-26. doi: 10.1016/j.jep.2014.01.006. Epub 2014 Jan 15. J Ethnopharmacol. 2014. PMID: 24440317
-
Saikosaponin D relieves unpredictable chronic mild stress induced depressive-like behavior in rats: involvement of HPA axis and hippocampal neurogenesis.Psychopharmacology (Berl). 2017 Nov;234(22):3385-3394. doi: 10.1007/s00213-017-4720-8. Epub 2017 Sep 5. Psychopharmacology (Berl). 2017. PMID: 28875366
-
Pharmacology, medical uses, and clinical translational challenges of Saikosaponin A: A review.Heliyon. 2024 Nov 15;10(22):e40427. doi: 10.1016/j.heliyon.2024.e40427. eCollection 2024 Nov 30. Heliyon. 2024. PMID: 39641011 Free PMC article. Review.
Cited by
-
Traditional Chinese Medicine in Depression Treatment: From Molecules to Systems.Front Pharmacol. 2020 May 7;11:586. doi: 10.3389/fphar.2020.00586. eCollection 2020. Front Pharmacol. 2020. PMID: 32457610 Free PMC article. Review.
-
Interpreting the Mechanism of Active Ingredients in Polygonati Rhizoma in Treating Depression by Combining Systemic Pharmacology and In Vitro Experiments.Nutrients. 2024 Apr 14;16(8):1167. doi: 10.3390/nu16081167. Nutrients. 2024. PMID: 38674858 Free PMC article.
-
Saikosaponin A alleviates depressive-like behavior induced by reserpine in mice by regulating gut microflora and inflammatory responses.PLoS One. 2025 Feb 10;20(2):e0311207. doi: 10.1371/journal.pone.0311207. eCollection 2025. PLoS One. 2025. PMID: 39928658 Free PMC article.
-
m6A Regulator-Mediated RNA Methylation Modification Patterns are Involved in the Pathogenesis and Immune Microenvironment of Depression.Front Genet. 2022 Apr 11;13:865695. doi: 10.3389/fgene.2022.865695. eCollection 2022. Front Genet. 2022. PMID: 35480327 Free PMC article.
-
Exploration of the mechanism by which icariin modulates hippocampal neurogenesis in a rat model of depression.Neural Regen Res. 2022 Mar;17(3):632-642. doi: 10.4103/1673-5374.320993. Neural Regen Res. 2022. PMID: 34380904 Free PMC article.
References
-
- Zalsman G, Braun M, Arendt M, et al. A comparison of the medical lethality of suicide attempts in bipolar and major depressive disorders. Bipolar Disord. 2006;8:558‐565. - PubMed
-
- Joca SR, Moreira FA, Wegener G. Atypical neurotransmitters and the neurobiology of depression. CNS Neurol Disord Drug Targets. 2015;14(8):1001‐1011. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

