The Chicken as a Model Organism to Study Heart Development
- PMID: 31767650
- PMCID: PMC7397825
- DOI: 10.1101/cshperspect.a037218
The Chicken as a Model Organism to Study Heart Development
Abstract
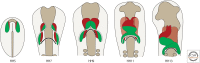
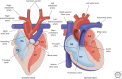
Heart development is a complex process and begins with the long-range migration of cardiac progenitor cells during gastrulation. This culminates in the formation of a simple contractile tube with multiple layers, which undergoes remodeling into a four-chambered heart. During this morphogenesis, additional cell populations become incorporated. It is important to unravel the underlying genetic and cellular mechanisms to be able to identify the embryonic origin of diseases, including congenital malformations, which impair cardiac function and may affect life expectancy or quality. Owing to the evolutionary conservation of development, observations made in nonamniote and amniote vertebrate species allow us to extrapolate to human. This review will focus on the contributions made to a better understanding of heart development through studying avian embryos-mainly the chicken but also quail embryos. We will illustrate the classic and recent approaches used in the avian system, give an overview of the important discoveries made, and summarize the early stages of cardiac development up to the establishment of the four-chambered heart.
Copyright © 2020 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures


Similar articles
-
Analysis of neural crest migration and differentiation by cross-species transplantation.J Vis Exp. 2012 Feb 7;(60):3622. doi: 10.3791/3622. J Vis Exp. 2012. PMID: 22349214 Free PMC article.
-
Embryonic Chicken (Gallus gallus domesticus) as a Model of Cardiac Biology and Development.Comp Med. 2019 May 1;69(3):184-203. doi: 10.30802/AALAS-CM-18-000061. Epub 2019 Jun 10. Comp Med. 2019. PMID: 31182184 Free PMC article. Review.
-
Dual labeling of neural crest cells and blood vessels within chicken embryos using Chick(GFP) neural tube grafting and carbocyanine dye DiI injection.J Vis Exp. 2015 May 28;(99):e52514. doi: 10.3791/52514. J Vis Exp. 2015. PMID: 26065540 Free PMC article.
-
Chick muscle development.Int J Dev Biol. 2018;62(1-2-3):127-136. doi: 10.1387/ijdb.170312cm. Int J Dev Biol. 2018. PMID: 29616720 Review.
-
Vagal neural crest cell migratory behavior: a transition between the cranial and trunk crest.Dev Dyn. 2011 Sep;240(9):2084-100. doi: 10.1002/dvdy.22715. Dev Dyn. 2011. PMID: 22016183 Free PMC article.
Cited by
-
Methodological and Ethical Considerations in the Use of Chordate Embryos in Biomedical Research.Int J Mol Sci. 2025 Mar 14;26(6):2624. doi: 10.3390/ijms26062624. Int J Mol Sci. 2025. PMID: 40141265 Free PMC article. Review.
-
SPADE: spatial deconvolution for domain specific cell-type estimation.Commun Biol. 2024 Apr 17;7(1):469. doi: 10.1038/s42003-024-06172-y. Commun Biol. 2024. PMID: 38632414 Free PMC article.
-
The avian embryo as a time-honoured animal model in developmental, biomedical and agricultural research.Philos Trans R Soc Lond B Biol Sci. 2025 Feb 27;380(1920):20230438. doi: 10.1098/rstb.2023.0438. Epub 2025 Feb 27. Philos Trans R Soc Lond B Biol Sci. 2025. PMID: 40010394 Review.
-
Xenopus: Experimental Access to Cardiovascular Development, Regeneration Discovery, and Cardiovascular Heart-Defect Modeling.Cold Spring Harb Perspect Biol. 2020 Jun 1;12(6):a037200. doi: 10.1101/cshperspect.a037200. Cold Spring Harb Perspect Biol. 2020. PMID: 31767648 Free PMC article. Review.
-
Particulate Matter 2.5 Induced Developmental Cardiotoxicity in Chicken Embryo and Hatchling.Front Pharmacol. 2020 Jun 5;11:841. doi: 10.3389/fphar.2020.00841. eCollection 2020. Front Pharmacol. 2020. PMID: 32581800 Free PMC article.
References
-
- Alsan BH, Schultheiss TM. 2002. Regulation of avian cardiogenesis by Fgf8 signaling. Development 129: 1935–1943. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
