Acetylcholine Mediates Dynamic Switching Between Information Coding Schemes in Neuronal Networks
- PMID: 31780905
- PMCID: PMC6861375
- DOI: 10.3389/fnsys.2019.00064
Acetylcholine Mediates Dynamic Switching Between Information Coding Schemes in Neuronal Networks
Abstract
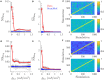
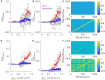
Rate coding and phase coding are the two major coding modes seen in the brain. For these two modes, network dynamics must either have a wide distribution of frequencies for rate coding, or a narrow one to achieve stability in phase dynamics for phase coding. Acetylcholine (ACh) is a potent regulator of neural excitability. Acting through the muscarinic receptor, ACh reduces the magnitude of the potassium M-current, a hyperpolarizing current that builds up as neurons fire. The M-current contributes to several excitability features of neurons, becoming a major player in facilitating the transition between Type 1 (integrator) and Type 2 (resonator) excitability. In this paper we argue that this transition enables a dynamic switch between rate coding and phase coding as levels of ACh release change. When a network is in a high ACh state variations in synaptic inputs will lead to a wider distribution of firing rates across the network and this distribution will reflect the network structure or pattern of external input to the network. When ACh is low, network frequencies become narrowly distributed and the structure of a network or pattern of external inputs will be represented through phase relationships between firing neurons. This work provides insights into how modulation of neuronal features influences network dynamics and information processing across brain states.
Keywords: acetylcholine; information coding; networks; neuromodulation; neuronal excitability.
Copyright © 2019 Roach, Eniwaye, Booth, Sander and Zochowski.
Figures
References
-
- Acebrón J. A., Bonilla L. L., Vicente C. J. P., Ritort F., Spigler R. (2005). The Kuramoto model: a simple paradigm for synchronization phenomena. Rev. Mod. Phys. 77, 137–185. 10.1103/RevModPhys.77.137 - DOI
LinkOut - more resources
Full Text Sources

