A WD40-Repeat Protein From the Recretohalophyte Limonium bicolor Enhances Trichome Formation and Salt Tolerance in Arabidopsis
- PMID: 31781150
- PMCID: PMC6861380
- DOI: 10.3389/fpls.2019.01456
A WD40-Repeat Protein From the Recretohalophyte Limonium bicolor Enhances Trichome Formation and Salt Tolerance in Arabidopsis
Abstract
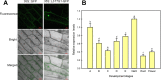
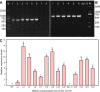
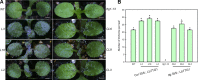
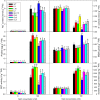
The Arabidopsis thaliana WD40-repeat protein TRANSPARENT TESTA GLABRA1 (TTG1) controls epidermis development, playing opposite roles in trichome differentiation and root hair formation. We isolated and characterized LbTTG1 (encoding a WD40-repeat protein with high sequence similarity to TTG1) from the recretohalophyte Limonium bicolor, which actively excretes absorbed salt via a salt gland. The complete open reading frame of LbTTG1 was 1,095 bp, encoding a protein of 364 amino acids, and showed highest expression during the salt gland initiation stage. We heterologously expressed LbTTG1 in wild type and ttg1-13 Arabidopsis plants to verify the protein's function, and the copies of LbTTG1 were identified in transgenic strains using southern blotting. Trichomes were extremely induced on the first true leaves of plants heterologously expressing LbTTG1, whereas no trichomes were produced by ttg1-13 plants. Conversely, plants heterologously expressing LbTTG1 produced fewer root hairs than ttg1-13 plants. In plants heterologously expressing LbTTG1 compared to controls, epidermis differentiation genes (GLABRA1 and GLABRA3) were up-regulated while genes encoding negative regulators of trichome development (TRIPTYCHON and CAPRICE) were down-regulated. Under increased NaCl concentrations, both of the transgenic lines showed enhanced germination and root length, and accumulated less malondialdehyde (MDA) and Na+ and produced more proline, soluble sugar, and higher glutathione S-transferase activity, compared with the ttg1-13 mutant. These results indicate that LbTTG1 participates in epidermis development in Arabidopsis, similarly to other WD40-repeat proteins, and specifically increases salt tolerance of transgenic Arabidopsis by reducing ion accumulation and increasing osmolyte levels.
Keywords: Arabidopsis; Limonium bicolor; WD40-repeat protein; heterologous expression; root hair; salt stress; trichome.
Copyright © 2019 Yuan, Leng, Zhang, Wang, Han and Wang.
Figures





Similar articles
-
Heterologous expression of the Limonium bicolor MYB transcription factor LbTRY in Arabidopsis thaliana increases salt sensitivity by modifying root hair development and osmotic homeostasis.Plant Sci. 2021 Jan;302:110704. doi: 10.1016/j.plantsci.2020.110704. Epub 2020 Oct 7. Plant Sci. 2021. PMID: 33288017
-
Importin-β From the Recretohalophyte Limonium bicolor Enhances Salt Tolerance in Arabidopsis thaliana by Reducing Root Hair Development and Abscisic Acid Sensitivity.Front Plant Sci. 2021 Jan 13;11:582459. doi: 10.3389/fpls.2020.582459. eCollection 2020. Front Plant Sci. 2021. PMID: 33519843 Free PMC article.
-
The genome of the recretohalophyte Limonium bicolor provides insights into salt gland development and salinity adaptation during terrestrial evolution.Mol Plant. 2022 Jun 6;15(6):1024-1044. doi: 10.1016/j.molp.2022.04.011. Epub 2022 May 5. Mol Plant. 2022. PMID: 35514085
-
Updates on molecular mechanisms in the development of branched trichome in Arabidopsis and nonbranched in cotton.Plant Biotechnol J. 2019 Sep;17(9):1706-1722. doi: 10.1111/pbi.13167. Epub 2019 Jun 11. Plant Biotechnol J. 2019. PMID: 31111642 Free PMC article. Review.
-
GLABRA2, A Common Regulator for Epidermal Cell Fate Determination and Anthocyanin Biosynthesis in Arabidopsis.Int J Mol Sci. 2019 Oct 9;20(20):4997. doi: 10.3390/ijms20204997. Int J Mol Sci. 2019. PMID: 31601032 Free PMC article. Review.
Cited by
-
Exogenous 6-BA enhances salt tolerance of Limonium bicolor by increasing the number of salt glands.Plant Cell Rep. 2023 Dec 23;43(1):12. doi: 10.1007/s00299-023-03104-8. Plant Cell Rep. 2023. PMID: 38135797
-
Genomic analysis of WD40 protein family in the mango reveals a TTG1 protein enhances root growth and abiotic tolerance in Arabidopsis.Sci Rep. 2021 Jan 26;11(1):2266. doi: 10.1038/s41598-021-81969-z. Sci Rep. 2021. PMID: 33500544 Free PMC article.
-
Alternaria alternata effector AaAlta1 targets CmWD40 and participates in regulating disease resistance in Chrysanthemum morifolium.PLoS Pathog. 2025 Mar 31;21(3):e1012942. doi: 10.1371/journal.ppat.1012942. eCollection 2025 Mar. PLoS Pathog. 2025. PMID: 40163540 Free PMC article.
-
An uncharacterized gene Lb1G04794 from Limonium bicolor promotes salt tolerance and trichome development in Arabidopsis.Front Plant Sci. 2022 Dec 8;13:1079534. doi: 10.3389/fpls.2022.1079534. eCollection 2022. Front Plant Sci. 2022. PMID: 36570955 Free PMC article.
-
Coupled Development of Salt Glands, Stomata, and Pavement Cells in Limonium bicolor.Front Plant Sci. 2021 Dec 9;12:745422. doi: 10.3389/fpls.2021.745422. eCollection 2021. Front Plant Sci. 2021. PMID: 34956255 Free PMC article.
References
-
- Ben-Simhon Z., Judeinstein S., Nadler-Hassar T., Trainin T., Bar-Ya’akov I., Borochov-Neori H., et al. (2011). A pomegranate (Punica granatum L.) WD40-repeat gene is a functional homologue of Arabidopsis TTG1 and is involved in the regulation of anthocyanin biosynthesis during pomegranate fruit development. Planta 234 (5), 865–881. 10.1007/s00425-011-1438-4 - DOI - PubMed
-
- Chen C., Yin S., Liu X., Liu B., Yang S., Xue S., et al. (2016). CsTTG1 Encodes a WD-repeat Protein That Regulates Fruit Wart Formation in Cucumis sativus through Interaction with the Homeodomain-leucine Zipper I Protein Mict. Plant Physiol. 171(2), 1156–1168. 10.1104/pp.16.00112 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

