IL-6 Negatively Regulates IL-22R α Expression on Epidermal Keratinocytes: Implications for Irritant Contact Dermatitis
- PMID: 31781680
- PMCID: PMC6875369
- DOI: 10.1155/2019/6276254
IL-6 Negatively Regulates IL-22R α Expression on Epidermal Keratinocytes: Implications for Irritant Contact Dermatitis
Abstract
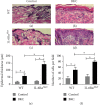
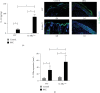
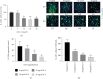
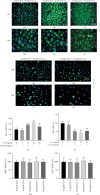
Irritant Contact Dermatitis (ICD) is characterized by epidermal hyperplasia and inflammatory cytokine release. IL-6 has been shown to be involved in the pathogenesis of ICD; however, the involvement of the IL-22/IL-22Rα axis and its relation to IL-6 in the inflammatory response following irritant exposure are unknown. Using a chemical model of ICD, it was observed that mice with a keratinocyte-specific knockout of IL-6Rα (IL-6Rα Δker) presented with increased inflammation and IL-22Rα and IL-22 protein expression relative to WT following irritant exposure, indicating that IL-6Rα deficiency in epidermal keratinocytes leads to the upregulation of IL-22Rα and its ligand during ICD. Furthermore, it was shown that IL-6 negatively regulates the expression of IL-22Rα on epidermal keratinocytes. This effect is functional as the effects of IL-22 on keratinocyte proliferation and differentiation were markedly reduced when keratinocytes were pretreated with IL-6 prior to IL-22 treatment. These results show that IL-6 modulates the IL-22/IL-22Rα axis in the skin and suggest that this occurrence may be associated with the increased epidermal hyperplasia and exacerbated inflammatory response observed in IL-6Rα Δker mice during ICD.
Copyright © 2019 Benjamin Frempah et al.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Keratinocyte-specific deletion of the IL-6RΑ exacerbates the inflammatory response during irritant contact dermatitis.Toxicology. 2019 Jul 1;423:123-131. doi: 10.1016/j.tox.2019.05.015. Epub 2019 May 31. Toxicology. 2019. PMID: 31158415
-
IL6Rα function in myeloid cells modulates the inflammatory response during irritant contact dermatitis.Exp Dermatol. 2019 Aug;28(8):948-955. doi: 10.1111/exd.13984. Epub 2019 Jul 3. Exp Dermatol. 2019. PMID: 31165501
-
Alloferon and IL-22 receptor expression regulation on the pathogenesis of imiquimod-induced psoriasis.Sci Rep. 2025 Feb 24;15(1):6671. doi: 10.1038/s41598-025-90961-w. Sci Rep. 2025. PMID: 39994364 Free PMC article.
-
Epidermal cytokines in murine cutaneous irritant responses.J Appl Toxicol. 2000 Jul-Aug;20(4):335-41. doi: 10.1002/1099-1263(200007/08)20:4<335::aid-jat698>3.0.co;2-l. J Appl Toxicol. 2000. PMID: 10942909 Review.
-
Irritant contact dermatitis: is there an immunologic component?Int Immunopharmacol. 2002 Feb;2(2-3):183-9. doi: 10.1016/s1567-5769(01)00171-0. Int Immunopharmacol. 2002. PMID: 11811923 Review.
Cited by
-
Exploring Causal Relationships Between Gut Microbiota, Inflammatory Cytokines, and Inflammatory Dermatoses: A Mendelian Randomization Study.Clin Cosmet Investig Dermatol. 2025 Mar 13;18:579-592. doi: 10.2147/CCID.S496091. eCollection 2025. Clin Cosmet Investig Dermatol. 2025. PMID: 40099043 Free PMC article.
-
FLASH Proton Pencil Beam Scanning Irradiation Minimizes Radiation-Induced Leg Contracture and Skin Toxicity in Mice.Cancers (Basel). 2021 Mar 1;13(5):1012. doi: 10.3390/cancers13051012. Cancers (Basel). 2021. PMID: 33804336 Free PMC article.
-
Therapeutic effect of bosentan on 2, 4-dinitrochlorobenzene (DNCB)-induced atopic dermatitis mouse model.Arch Dermatol Res. 2025 Feb 18;317(1):436. doi: 10.1007/s00403-025-03955-z. Arch Dermatol Res. 2025. PMID: 39966154
-
IL-22 in Atopic Dermatitis.Cells. 2024 Aug 22;13(16):1398. doi: 10.3390/cells13161398. Cells. 2024. PMID: 39195286 Free PMC article. Review.
-
Veronica persica Ethanol Extract Ameliorates Dinitrochlorobenzene-Induced Atopic Dermatitis-like Skin Inflammation in Mice, Likely by Inducing Nrf2/HO-1 Signaling.Antioxidants (Basel). 2023 Jun 13;12(6):1267. doi: 10.3390/antiox12061267. Antioxidants (Basel). 2023. PMID: 37371997 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

