Regional differences in Alzheimer's disease pathology confound behavioural rescue after amyloid-β attenuation
- PMID: 31782760
- PMCID: PMC6935751
- DOI: 10.1093/brain/awz371
Regional differences in Alzheimer's disease pathology confound behavioural rescue after amyloid-β attenuation
Erratum in
-
Corrigendum.Brain. 2020 Mar 1;143(3):e23. doi: 10.1093/brain/awz409. Brain. 2020. PMID: 31891369 Free PMC article. No abstract available.
-
Erratum.Brain. 2020 Mar 1;143(3):e24. doi: 10.1093/brain/awaa007. Brain. 2020. PMID: 32333675 Free PMC article. No abstract available.
Abstract
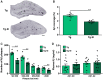
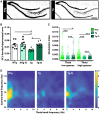
Failure of Alzheimer's disease clinical trials to improve or stabilize cognition has led to the need for a better understanding of the driving forces behind cognitive decline in the presence of active disease processes. To dissect contributions of individual pathologies to cognitive function, we used the TgF344-AD rat model, which recapitulates the salient hallmarks of Alzheimer's disease pathology observed in patient populations (amyloid, tau inclusions, frank neuronal loss, and cognitive deficits). scyllo-Inositol treatment attenuated amyloid-β peptide in disease-bearing TgF344-AD rats, which rescued pattern separation in the novel object recognition task and executive function in the reversal learning phase of the Barnes maze. Interestingly, neither activities of daily living in the burrowing task nor spatial memory in the Barnes maze were rescued by attenuating amyloid-β peptide. To understand the pathological correlates leading to behavioural rescue, we examined the neuropathology and in vivo electrophysiological signature of the hippocampus. Amyloid-β peptide attenuation reduced hippocampal tau pathology and rescued adult hippocampal neurogenesis and neuronal function, via improvements in cross-frequency coupling between theta and gamma bands. To investigate mechanisms underlying the persistence of spatial memory deficits, we next examined neuropathology in the entorhinal cortex, a region whose input to the hippocampus is required for spatial memory. Reduction of amyloid-β peptide in the entorhinal cortex had no effect on entorhinal tau pathology or entorhinal-hippocampal neuronal network dysfunction, as measured by an impairment in hippocampal response to entorhinal stimulation. Thus, rescue or not of cognitive function is dependent on regional differences of amyloid-β, tau and neuronal network dysfunction, demonstrating the importance of staging disease in patients prior to enrolment in clinical trials. These results further emphasize the need for combination therapeutic approaches across disease progression.
Keywords: Alzheimer’s disease; amyloid-β; cognition; hippocampal-entorhinal circuitry; tau.
© The Author(s) (2019). Published by Oxford University Press on behalf of the Guarantors of Brain.
Figures




References
-
- Anckaerts C, Blockx I, Summer P. Early functional connectivity deficits and progressive microstructural alterations in the TgF344-AD rat model of Alzheimer’s disease: a longitudinal MRI study. Neurobiol Dis 2019; 124: 93–107. - PubMed
-
- Bazzigaluppi P, Beckett TL, Koletar MM, Lai AY, Joo IL, Brown ME, et al.Early-stage attenuation of phase-amplitude coupling in the hippocampus and medial prefrontal cortex in a transgenic rat model of Alzheimer’s disease. J Neurochem 2018; 144: 669–79. - PubMed

