A mouse model for vitamin D-induced human cathelicidin antimicrobial peptide gene expression
- PMID: 31783153
- PMCID: PMC7089838
- DOI: 10.1016/j.jsbmb.2019.105552
A mouse model for vitamin D-induced human cathelicidin antimicrobial peptide gene expression
Abstract
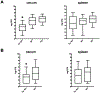
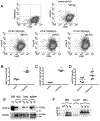
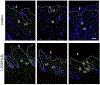
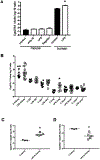
In humans and other primates, 1,25(OH)2vitamin D3 regulates the expression of the cathelicidin antimicrobial peptide (CAMP) gene via toll-like receptor (TLR) signaling that activates the vitamin D pathway. Mice and other mammals lack the vitamin D response element (VDRE) in their CAMP promoters. To elucidate the biological importance of this pathway, we generated transgenic mice that carry a genomic DNA fragment encompassing the entire human CAMP gene and crossed them with Camp knockout (KO) mice. We observed expression of the human transgene in various tissues and innate immune cells. However, in mouse CAMP transgenic macrophages, TLR activation in the presence of 25(OH)D3 did not induce expression of either CAMP or CYP27B1 as would normally occur in human macrophages, reinforcing important species differences in the actions of vitamin D. Transgenic mice did show increased resistance to colonization by Salmonella typhimurium in the gut. Furthermore, the human CAMP gene restored wound healing in the skin of Camp KO mice. Topical application of 1,25(OH)2vitamin D3 to the skin of CAMP transgenic mice induced CAMP expression and increased killing of Staphylococcus aureus in a wound infection model. Our model can help elucidate the biological importance of the vitamin D-cathelicidin pathway in both pathogenic and non-pathogenic states.
Keywords: Cathelicidin; Cyp27b1; Innate immunity; Macrophage; TLR; Vitamin D.
Copyright © 2019 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare no conflicts of interest.
Figures




References
-
- Wang TT, Nestel FP, Bourdeau V, Nagai Y, Wang Q, Liao J, Tavera-Mendoza L, Lin R, Hanrahan JW, Mader S, and White JH (2004) Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J Immunol 173, 2909–2912 - PubMed
-
- Gombart AF, Borregaard N, and Koeffler HP (2005) Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3. FASEB J 19, 1067–1077 - PubMed
-
- Weber G, Heilborn JD, Chamorro Jimenez CI, Hammarsjo A, Torma H, and Stahle M (2005) Vitamin D induces the antimicrobial protein hCAP18 in human skin. J Invest Dermatol 124, 1080–1082 - PubMed
-
- Zanetti M, Gennaro R, and Romeo D (1995) Cathelicidins: a novel protein family with a common proregion and a variable C-terminal antimicrobial domain. FEBS Lett 374, 1–5 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

