Visualizing bovine leukemia virus (BLV)-infected cells and measuring BLV proviral loads in the milk of BLV seropositive dams
- PMID: 31783914
- PMCID: PMC6884895
- DOI: 10.1186/s13567-019-0724-1
Visualizing bovine leukemia virus (BLV)-infected cells and measuring BLV proviral loads in the milk of BLV seropositive dams
Abstract
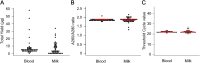
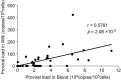
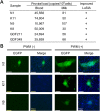
Bovine leukemia virus (BLV) infects cattle and causes serious problems for the cattle industry, worldwide. Vertical transmission of BLV occurs via in utero infection and ingestion of infected milk and colostrum. The aim of this study was to clarify whether milk is a risk factor in BLV transmission by quantifying proviral loads in milk and visualizing the infectivity of milk. We collected blood and milk from 48 dams (46 BLV seropositive dams and 2 seronegative dams) from seven farms in Japan and detected the BLV provirus in 43 blood samples (89.6%) but only 22 milk samples (45.8%) using BLV-CoCoMo-qPCR-2. Although the proviral loads in the milk tended to be lower, a positive correlation was firstly found between the proviral loads with blood and milk. Furthermore, the infectivity of milk cells with BLV was visualized ex vivo using a luminescence syncytium induction assay (LuSIA) based on CC81-GREMG cells, which form syncytia expressing enhanced green fluorescent protein (EGFP) in response to BLV Tax and Env expressions when co-cultured with BLV-infected cells. Interestingly, in addition to one BLV-infected dam with lymphoma, syncytia with EGFP fluorescence were observed in milk cells from six BLV-infected, but healthy, dams by an improved LuSIA, which was optimized for milk cells. This is the first report demonstrating the infectious capacity of cells in milk from BLV-infected dams by visualization of BLV infection ex vivo. Thus, our results suggest that milk is a potential risk factor for BLV vertical spread through cell to cell transmission.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
A sensitive luminescence syncytium induction assay (LuSIA) based on a reporter plasmid containing a mutation in the glucocorticoid response element in the long terminal repeat U3 region of bovine leukemia virus.Virol J. 2019 May 20;16(1):66. doi: 10.1186/s12985-019-1172-2. Virol J. 2019. PMID: 31109347 Free PMC article.
-
Risk factors associated with increased bovine leukemia virus proviral load in infected cattle in Japan from 2012 to 2014.Virus Res. 2015 Dec 2;210:283-90. doi: 10.1016/j.virusres.2015.08.020. Epub 2015 Aug 29. Virus Res. 2015. PMID: 26321160
-
Estimation of bovine leukemia virus (BLV) proviral load harbored by lymphocyte subpopulations in BLV-infected cattle at the subclinical stage of enzootic bovine leucosis using BLV-CoCoMo-qPCR.BMC Vet Res. 2013 May 4;9:95. doi: 10.1186/1746-6148-9-95. BMC Vet Res. 2013. PMID: 23641811 Free PMC article.
-
Invited review: Bovine leukemia virus-Transmission, control, and eradication.J Dairy Sci. 2021 Jun;104(6):6358-6375. doi: 10.3168/jds.2020-18925. Epub 2021 Mar 23. J Dairy Sci. 2021. PMID: 33741150 Review.
-
Bovine Leukemia Virus Infection in Neonatal Calves. Risk Factors and Control Measures.Front Vet Sci. 2018 Oct 25;5:267. doi: 10.3389/fvets.2018.00267. eCollection 2018. Front Vet Sci. 2018. PMID: 30410920 Free PMC article. Review.
Cited by
-
BoLA-DRB3 Polymorphism Associated with Bovine Leukemia Virus Infection and Proviral Load in Holstein Cattle in Egypt.Pathogens. 2023 Dec 14;12(12):1451. doi: 10.3390/pathogens12121451. Pathogens. 2023. PMID: 38133334 Free PMC article.
-
Correlation between the Biodistribution of Bovine Leukemia Virus in the Organs and the Proviral Load in the Peripheral Blood during Early Stages of Experimentally Infected Cattle.Pathogens. 2023 Jan 12;12(1):130. doi: 10.3390/pathogens12010130. Pathogens. 2023. PMID: 36678478 Free PMC article.
-
PRMT5 Is Required for Bovine Leukemia Virus Infection In Vivo and Regulates BLV Gene Expression, Syncytium Formation, and Glycosylation In Vitro.Viruses. 2020 Jun 16;12(6):650. doi: 10.3390/v12060650. Viruses. 2020. PMID: 32560231 Free PMC article.
-
Bovine leukemia viral DNA found on human breast tissue is genetically related to the cattle virus.One Health. 2021 Apr 20;13:100252. doi: 10.1016/j.onehlt.2021.100252. eCollection 2021 Dec. One Health. 2021. PMID: 33997236 Free PMC article.
-
Effect of formic acid treatment on colostrum quality, and on absorption and function of immunoglobulins: a randomized controlled trial in Holstein dairy calves.BMC Vet Res. 2022 Aug 17;18(1):318. doi: 10.1186/s12917-022-03418-x. BMC Vet Res. 2022. PMID: 35978339 Free PMC article.
References
-
- Gillet N, Florins A, Boxus M, Burteau C, Nigro A, Vandermeers F, Balon H, Bouzar AB, Defoiche J, Burny A, Reichert M, Kettmann R, Willems L. Mechanisms of leukemogenesis induced by bovine leukemia virus: prospects for novel anti-retroviral therapies in human. Retrovirology. 2007;4:18. doi: 10.1186/1742-4690-4-18. - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

