Lithium Perchlorate-, Acetic Anhydride-, and Triphenylphosphine-assisted Multicomponent Syntheses of 4-Unsubstituted 2,5-Dioxooctahydroquinoline-3-carboxylates and 3-carbonitriles
- PMID: 31787785
- PMCID: PMC6884338
- DOI: 10.1016/j.tet.2013.08.081
Lithium Perchlorate-, Acetic Anhydride-, and Triphenylphosphine-assisted Multicomponent Syntheses of 4-Unsubstituted 2,5-Dioxooctahydroquinoline-3-carboxylates and 3-carbonitriles
Abstract
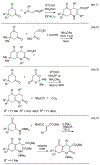
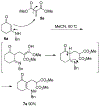
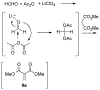
Lithium perchlorate and acetic anhydride were the key additives for the multi-component reaction between 3-aminocyclohex-2-enones, formaldehyde, and malonates yielding adducts that were annulated under acidic conditions to afford bicyclic 2,5-dioxooctahydroquinoline-3-carboxylates. When methyl cyanoacetate was subjected to the same reaction conditions in the presence of a catalytic amount of triphenylphosphine, the bicyclic 2,5-dioxooctahydroquinoline-3-carbonitriles were obtained in a one-flask reaction.
Keywords: 2,5-dioxooctahydroquinolines; Multicomponent reaction; acetic anhydride; catalysis; lithium perchlorate; triphenylphosphine.
Figures

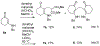
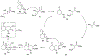
Similar articles
-
Pd(II)-catalyzed regio-, enantio-, and diastereoselective 1,4-addition of azlactones formed in situ from racemic unprotected amino acids and acetic anhydride.J Org Chem. 2012 Dec 7;77(23):10846-55. doi: 10.1021/jo302177t. Epub 2012 Nov 29. J Org Chem. 2012. PMID: 23193999
-
Chromogenic reactions of tertiary amines with polycarboxylic acids and acetic anhydride: carbon suboxide as the reactive species in the malonic acid reagent.J Pharm Sci. 1987 Oct;76(10):834-8. doi: 10.1002/jps.2600761018. J Pharm Sci. 1987. PMID: 3430350
-
Asymmetric Michael addition of malonates to enones catalyzed by a primary β-amino acid and its lithium salt.J Org Chem. 2011 Oct 21;76(20):8513-7. doi: 10.1021/jo201429w. Epub 2011 Sep 16. J Org Chem. 2011. PMID: 21894973
-
Copper-catalyzed Perkin-acyl-Mannich reaction of acetic anhydride with pyridine: expeditious entry to unconventional piperidines.Org Lett. 2011 Oct 7;13(19):5152-5. doi: 10.1021/ol202027k. Epub 2011 Sep 6. Org Lett. 2011. PMID: 21894883
-
Derivatization with acetic anhydride: applications to the analysis of biogenic amines and psychiatric drugs by gas chromatography and mass spectrometry.J Pharmacol Toxicol Methods. 1994 Jun;31(3):141-8. doi: 10.1016/1056-8719(94)90076-0. J Pharmacol Toxicol Methods. 1994. PMID: 8068975 Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources
