Involvement of Phosphatase and Tensin Homolog in Cyclin-Dependent Kinase 4/6 Inhibitor-Induced Blockade of Glioblastoma
- PMID: 31787897
- PMCID: PMC6854038
- DOI: 10.3389/fphar.2019.01316
Involvement of Phosphatase and Tensin Homolog in Cyclin-Dependent Kinase 4/6 Inhibitor-Induced Blockade of Glioblastoma
Abstract
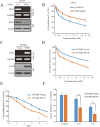
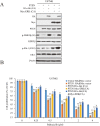
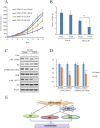
Dysregulation of retinoblastoma (Rb) signaling pathway have been established as a requirement for glioblastoma (GBM) initiation and progression, which suggests that blockade of CDK4/6-Rb signaling axis for GBM treatment. Palbociclib, a selective inhibitor of the cyclin-dependent kinases CDK4/6, has been applied for breast cancer treatment. However, its efficacy against glioblastoma has not been well clarified. Here, effects of CDK4/6 inhibitors on various kinds of GBM cell lines are investigated and the functional mechanisms are identified. Data showed that cells with diverse PTEN status respond to palbociclib differently. Gain-of-function and loss-of-function studies indicated that PTEN enhanced the sensitivity of GBM cells to palbociclib in vitro and in vivo, which was associated with suppressions of Akt and ERK signaling and independent of Rb signaling inhibition. Hence, our findings support that palbociclib selectively.
Keywords: CDK4/6 inhibitor; PTEN; glioblastoma; palbociclib; sensitivity.
Copyright © 2019 Liu, Yuan, Li, Qi, Guo, Yang, Zhou, Xu, Chen, Yang, Liu, Li, Yao and Jiang.
Figures



References
-
- Bollard J., Miguela V., Ruiz de Galarreta M., Venkatesh A., Bian C. B., Roberto M. P., et al. (2016). Palbociclib (PD-0332991), a selective CDK4/6 inhibitor, restricts tumour growth in preclinical models of hepatocellular carcinoma. Gut. 66, 1286–1296. 10.1136/gutjnl-2016-312268 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

