A review of the WHO malaria rapid diagnostic test product testing programme (2008-2018): performance, procurement and policy
- PMID: 31791354
- PMCID: PMC6889598
- DOI: 10.1186/s12936-019-3028-z
A review of the WHO malaria rapid diagnostic test product testing programme (2008-2018): performance, procurement and policy
Abstract
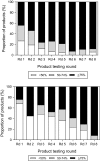
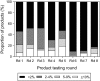
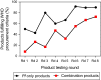
Malaria rapid diagnostic tests (RDTs) emerged in the early 1990s into largely unregulated markets, and uncertain field performance was a major concern for the acceptance of tests for malaria case management. This, combined with the need to guide procurement decisions of UN agencies and WHO Member States, led to the creation of an independent, internationally coordinated RDT evaluation programme aiming to provide comparative performance data of commercially available RDTs. Products were assessed against Plasmodium falciparum and Plasmodium vivax samples diluted to two densities, along with malaria-negative samples from healthy individuals, and from people with immunological abnormalities or non-malarial infections. Three measures were established as indicators of performance, (i) panel detection score (PDS) determined against low density panels prepared from P. falciparum and P. vivax wild-type samples, (ii) false positive rate, and (iii) invalid rate, and minimum criteria defined. Over eight rounds of the programme, 332 products were tested. Between Rounds 1 and 8, substantial improvements were seen in all performance measures. The number of products meeting all criteria increased from 26.8% (11/41) in Round 1, to 79.4% (27/34) in Round 8. While products submitted to further evaluation rounds under compulsory re-testing did not show improvement, those voluntarily resubmitted showed significant increases in P. falciparum (p = 0.002) and P. vivax PDS (p < 0.001), with more products meeting the criteria upon re-testing. Through this programme, the differentiation of products based on comparative performance, combined with policy changes has been influential in the acceptance of malaria RDTs as a case-management tool, enabling a policy of parasite-based diagnosis prior to treatment. Publication of product testing results has produced a transparent market allowing users and procurers to clearly identify appropriate products for their situation, and could form a model for introduction of other, broad-scale diagnostics.
Keywords: Malaria; Plasmodium falciparum; Plasmodium vivax; Product improvement; Rapid diagnostic tests.
Conflict of interest statement
JB and CK, declare no competing interests. SJ reports payment from WHO for writing the manuscript. MG reports agreements with WHO and FIND for performance of work to conduct statistical analysis. QC reports agreements with WHO and FIND for performance of work to characterize the parasite panel. SN, DM, WO and JL report payment from FIND for sample collection and characterization. JG reports payment of salary through a contract with FIND. JC reports payment of salary from WHO. IG and SI report grants with the Bill and Melinda Gates Foundation, UNITAID, UK Department for International Development, Australian Agency for International Development and USAID during the conduct of the study. PC and RRC report grants with WHO and FIND during the conduct of the study. DB reports past grants from The Bill and Melinda Gates Foundation and UNITAID during the conduct of the study.
Figures

References
-
- WHO . World malaria report. Geneva: World Health Organization; 2018.
-
- WHO. New perspectives: malaria diagnosis. Report of joint WHO/USAID informal consultation 25–27 October. Geneva: World Health Organization; 2000.
-
- Thepsamarn P, Prayoollawongsa N, Puksupa P, Puttoom P, Thaidumrong P, Wongchai S, et al. The ICT malaria Pf: a simple, rapid dipstick test for the diagnosis of Plasmodium falciparum malaria at the Thai-Myanmar border. Southeast Asian J Trop Med Public Health. 1997;28:723–726. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

