Clinical safety of ProMRI implantable cardioverter-defibrillator systems during head and lower lumbar magnetic resonance imaging at 1.5 Tesla
- PMID: 31796767
- PMCID: PMC6890633
- DOI: 10.1038/s41598-019-54342-4
Clinical safety of ProMRI implantable cardioverter-defibrillator systems during head and lower lumbar magnetic resonance imaging at 1.5 Tesla
Abstract
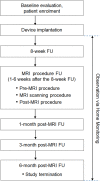
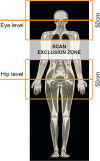
Magnetic resonance imaging (MRI) has long been contraindicated in patients with implanted pacemakers, defibrillators, and cardiac resynchronisation therapy (CRT) devices due to the risk of adverse effects through electromagnetic interference. Since many recipients of these devices will have a lifetime indication for an MRI scan, the implantable systems should be developed as 'MRI-conditional' (be safe for the MRI environment under predefined conditions). We evaluated the clinical safety of several Biotronik ProMRI ('MRI-conditional') defibrillator and CRT systems during head and lower lumbar MRI scans at 1.5 Tesla. The study enrolled 194 patients at 22 sites in Australia, Canada, and Europe. At ≥9 weeks after device implantation, predefined, non-diagnostic, specific absorption rate (SAR)-intensive head and lower lumbar MRI scans (total ≈30 minutes per patient) were performed in 146 patients that fulfilled pre-procedure criteria. Three primary endpoints were evaluated: freedom from serious adverse device effects (SADEs) related to MRI and defibrillator/CRT (leading to death, hospitalisation, life-threatening condition, or potentially requiring implanted system revision or replacement), pacing threshold increase, and sensing amplitude decrease, all at the 1-month post-MRI clinical visit. No MRI-related SADE occurred. Lead values remained stable, measured in clinic and monitored daily by the manufacturer home monitoring technology.
Conflict of interest statement
W.B. is scientific advisor of Biotronik. D.H.L. is supported by a Robert J. Craig Lectureship from the University of Adelaide and received lecture and/or consulting fees from St Jude Medical, Boehringer Ingelheim, Bayer, and Pfizer. A.McG. received support from Biotronik to attend past meetings (travel and accommodation). S.F. is employee of Biotronik. Other authors declare no potential conflict of interest.
Figures
Similar articles
-
Clinical safety of the ProMRI implantable cardioverter-defibrillator systems during head and lower lumbar magnetic resonance imaging at 3 T: results of the ProMRI 3T ENHANCED Master study.Europace. 2019 Nov 1;21(11):1678-1685. doi: 10.1093/europace/euz189. Europace. 2019. PMID: 31322701 Free PMC article.
-
Clinical safety of the ProMRI pacemaker system in patients subjected to head and lower lumbar 1.5-T magnetic resonance imaging scanning conditions.Heart Rhythm. 2015 Jun;12(6):1183-91. doi: 10.1016/j.hrthm.2015.02.010. Epub 2015 Feb 11. Heart Rhythm. 2015. PMID: 25680307
-
Clinical safety of an MRI conditional implantable cardioverter defibrillator system: A prospective Monocenter ICD-Magnetic resonance Imaging feasibility study (MIMI).J Magn Reson Imaging. 2016 Mar;43(3):574-84. doi: 10.1002/jmri.25037. Epub 2015 Sep 3. J Magn Reson Imaging. 2016. PMID: 26335332
-
Implications of pacemakers and implantable cardioverter defibrillators in urological practice.J Urol. 2011 Oct;186(4):1198-205. doi: 10.1016/j.juro.2011.02.2697. Epub 2011 Aug 16. J Urol. 2011. PMID: 21849189 Review.
-
MRI and implantable cardiac electronic devices.Curr Opin Cardiol. 2015 Jan;30(1):65-73. doi: 10.1097/HCO.0000000000000132. Curr Opin Cardiol. 2015. PMID: 25469592 Review.
Cited by
-
Utilization and programming of an automatic MRI recognition feature for cardiac rhythm management devices.Heart Rhythm O2. 2021 Mar 9;2(2):132-137. doi: 10.1016/j.hroo.2021.03.002. eCollection 2021 Apr. Heart Rhythm O2. 2021. PMID: 34113915 Free PMC article.
-
Magnetic resonance imaging in patients with cardiac implantable electronic devices: the RESONANCE Spanish registry.Europace. 2024 Nov 1;26(11):euae277. doi: 10.1093/europace/euae277. Europace. 2024. PMID: 39498837 Free PMC article.
-
Is diversity harmful?-Mixed-brand cardiac implantable electronic devices undergoing magnetic resonance imaging.Wien Klin Wochenschr. 2022 Apr;134(7-8):286-293. doi: 10.1007/s00508-021-01924-w. Epub 2021 Aug 17. Wien Klin Wochenschr. 2022. PMID: 34402991 Free PMC article.
-
Cold atmospheric plasma stabilizes mismatch repair for effective, uniform treatment of diverse colorectal cancer cell types.Sci Rep. 2024 Feb 13;14(1):3599. doi: 10.1038/s41598-024-54020-0. Sci Rep. 2024. PMID: 38351129 Free PMC article.
-
Signal voids of active cardiac implants at 3.0 T CMR.Sci Rep. 2022 Apr 15;12(1):6285. doi: 10.1038/s41598-022-09690-z. Sci Rep. 2022. PMID: 35428775 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

