Acute Cortical Lesions in MELAS Syndrome: Anatomic Distribution, Symmetry, and Evolution
- PMID: 31806591
- PMCID: PMC6975311
- DOI: 10.3174/ajnr.A6325
Acute Cortical Lesions in MELAS Syndrome: Anatomic Distribution, Symmetry, and Evolution
Abstract
Background and purpose: Mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS) syndrome is a rare mitochondrial disorder affecting children and young adults. Stroke-like episodes are often associated with acute cortical lesions in the posterior cerebral cortex and are classically described as asymmetric and transient. In this study we assessed the anatomic distribution of acute cortical lesions, the incidence of symmetry, and the temporal evolution of lesions.
Materials and methods: This was a retrospective cohort study of patients who had a confirmed genetic diagnosis of a pathogenic variant associated with MELAS and MR imaging performed at our center (2006-2018). Each MR imaging study was assessed for new lesions using T1, T2, FLAIR, DWI, ADC, and SWI. The anatomic location, symmetry, and temporal evolution of lesions were analyzed.
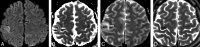
Results: Eight patients with the same pathogenic variant of MELAS (MT-TL1 m.3243A>G) with 31 MR imaging studies were included. Forty-one new lesions were identified in 17 of the studies (5 deep, 36 cortical). Cortical lesions most commonly affected the primary visual cortex, the middle-third of the primary somatosensory cortex, and the primary auditory cortex. Thirty of 36 cortical lesions had acute cortical diffusion restriction, of which 21 developed cortical laminar necrosis on subacute imaging. Six of 11 studies with multiple lesions showed symmetric cortical involvement.
Conclusions: Acute cortical lesions in MELAS most commonly affect the primary visual, somatosensory, and auditory cortices, all regions of high neuronal density and metabolic demand. The most common pattern of temporal evolution is acute cortical diffusion restriction with subacute cortical laminar necrosis and chronic volume loss. Symmetric involvement is more common than previously described.
© 2020 by American Journal of Neuroradiology.
Figures

Comment in
-
Stroke-like Episodes in m.3243A≥G Carriers Need to Be Monitored by MRI Starting with the Onset of Clinical Manifestations.AJNR Am J Neuroradiol. 2020 Mar;41(3):E17-E18. doi: 10.3174/ajnr.A6418. Epub 2020 Feb 6. AJNR Am J Neuroradiol. 2020. PMID: 32029472 Free PMC article. No abstract available.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
