Durable multitransgene expression in vivo using systemic, nonviral DNA delivery
- PMID: 31807699
- PMCID: PMC6881169
- DOI: 10.1126/sciadv.aax0217
Durable multitransgene expression in vivo using systemic, nonviral DNA delivery
Abstract
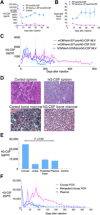
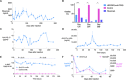
Recombinant adeno-associated virus (AAV) vectors are transforming therapies for rare human monogenic deficiency diseases. However, adaptive immune responses to AAV and its limited DNA insert capacity, restrict their therapeutic potential. HEDGES (high-level extended duration gene expression system), a nonviral DNA- and liposome-based gene delivery platform, overcomes these limitations in immunocompetent mice. Specifically, one systemic HEDGES injection durably produces therapeutic levels of transgene-encoded human proteins, including FDA-approved cytokines and monoclonal antibodies, without detectable integration into genomic DNA. HEDGES also controls protein production duration from <3 weeks to >1.5 years, does not induce anti-vector immune responses, is reexpressed for prolonged periods following reinjection, and produces only transient minimal toxicity. HEDGES can produce extended therapeutic levels of multiple transgene-encoded therapeutic human proteins from DNA inserts >1.5-fold larger than AAV-based therapeutics, thus creating combinatorial interventions to effectively treat common polygenic diseases driven by multigenic abnormalities.
Copyright © 2019 The Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science. No claim to original U.S. Government Works. Distributed under a Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC).
Figures

Similar articles
-
Adeno-associated virus (AAV) vectors achieve prolonged transgene expression in mouse myocardium and arteries in vivo: a comparative study with adenovirus vectors.Int J Cardiol. 2003 Aug;90(2-3):229-38. doi: 10.1016/s0167-5273(02)00554-5. Int J Cardiol. 2003. PMID: 12957756
-
Gene transfer into hepatocytes mediated by helper virus-free HSV/AAV hybrid vectors.Mol Med. 1997 Dec;3(12):813-25. Mol Med. 1997. PMID: 9440115 Free PMC article.
-
Expression of therapeutic levels of factor VIII in hemophilia A mice using a novel adeno/adeno-associated hybrid virus.Thromb Haemost. 2004 Aug;92(2):317-27. doi: 10.1160/TH04-02-0068. Thromb Haemost. 2004. PMID: 15269828
-
Expressing Transgenes That Exceed the Packaging Capacity of Adeno-Associated Virus Capsids.Hum Gene Ther Methods. 2016 Feb;27(1):1-12. doi: 10.1089/hgtb.2015.140. Hum Gene Ther Methods. 2016. PMID: 26757051 Free PMC article. Review.
-
Current development of nonviral-mediated gene transfer.Drug News Perspect. 2007 May;20(4):227-31. doi: 10.1358/dnp.2007.20.4.1103528. Drug News Perspect. 2007. PMID: 17637935 Review.
Cited by
-
The hybrid lipoplex induces cytoskeletal rearrangement via autophagy/RhoA signaling pathway for enhanced anticancer gene therapy.Nat Commun. 2025 Jan 2;16(1):339. doi: 10.1038/s41467-024-55727-4. Nat Commun. 2025. PMID: 39747218 Free PMC article.
-
Multi-step screening of DNA/lipid nanoparticles and co-delivery with siRNA to enhance and prolong gene expression.Nat Commun. 2022 Jul 25;13(1):4282. doi: 10.1038/s41467-022-31993-y. Nat Commun. 2022. PMID: 35879315 Free PMC article.
-
Advancing CRISPR-Based Solutions for COVID-19 Diagnosis and Therapeutics.Cells. 2024 Oct 30;13(21):1794. doi: 10.3390/cells13211794. Cells. 2024. PMID: 39513901 Free PMC article. Review.
-
CRISPR use in diagnosis and therapy for COVID-19.Methods Microbiol. 2022;50:123-150. doi: 10.1016/bs.mim.2022.03.002. Epub 2022 May 6. Methods Microbiol. 2022. PMID: 38013928 Free PMC article.
-
An overview of basic molecular biology of SARS-CoV-2 and current COVID-19 prevention strategies.Gene Rep. 2021 Jun;23:101122. doi: 10.1016/j.genrep.2021.101122. Epub 2021 Apr 1. Gene Rep. 2021. PMID: 33821222 Free PMC article. Review.
References
-
- Templeton N. S., Cationic liposome-mediated gene delivery in vivo. Biosci. Rep. 22, 283–295 (2002). - PubMed
-
- Yew N. S., Zhao H., Wu I. H., Song A., Tousignant J. D., Przybylska M., Cheng S. H., Reduced inflammatory response to plasmid DNA vectors by elimination and inhibition of immunostimulatory cpg motifs. Mol. Ther. 1, 255–262 (2000). - PubMed
-
- Tu G., Kirchmaier A. L., Liggitt D., Liu Y., Liu S., Yu W. H., Heath T. D., Thor A., Debs R. J., Non-replicating epstein-barr virus-based plasmids extend gene expression and can improve gene therapy in vivo. J. Biol. Chem. 275, 30408–30416 (2000). - PubMed
-
- Liu Y., Liggitt D., Zhong W., Tu G., Gaensler K., Debs R., Cationic liposome-mediated intravenous gene delivery. J. Biol. Chem. 270, 24864–24870 (1995). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

