Inflammatory Pathways Following Subarachnoid Hemorrhage
- PMID: 31808009
- PMCID: PMC11448815
- DOI: 10.1007/s10571-019-00767-4
Inflammatory Pathways Following Subarachnoid Hemorrhage
Abstract
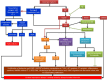
Aneurysmal subarachnoid hemorrhage (SAH) is an acute cerebrovascular emergency resulting from the rupture of a brain aneurysm. Despite only accounting for 5% of all strokes, SAH imposes a significant health burden on society due to its relatively young age at onset. Those who survive the initial bleed are often afflicted with severe disabilities thought to result from delayed cerebral ischemia (DCI). Consequently, elucidating the underlying mechanistic pathways implicated in DCI development following SAH remains a priority. Neuroinflammation has recently been implicated as a promising new theory for the development of SAH complications. However, despite this interest, clinical trials have failed to provide consistent evidence for the use of anti-inflammatory agents in SAH patients. This may be explained by the complexity of SAH as a plethora of inflammatory pathways have been shown to be activated in the disease. By determining how these pathways may overlap and interact, we hope to better understand the developmental processes of SAH complications and how to prevent them. The goal of this review is to provide insight into the available evidence regarding the molecular pathways involved in the development of inflammation following SAH and how SAH complications may arise as a result of these inflammatory pathways.
Keywords: Delayed cerebral ischemia; Inflammation; Ruptured aneurysm; Subarachnoid hemorrhage; Vasospasm.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
References
-
- Ansar S, Vikman P, Nielsen M, Edvinsson L (2007) Cerebrovascular ETB, 5-HT1B, and AT1 receptor upregulation correlates with reduction in regional CBF after subarachnoid hemorrhage. Am J Physiol 293:3750 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

