Heterogeneity Profoundly Alters Emergent Stress Fields in Constrained Multicellular Systems
- PMID: 31812354
- PMCID: PMC6950641
- DOI: 10.1016/j.bpj.2019.11.018
Heterogeneity Profoundly Alters Emergent Stress Fields in Constrained Multicellular Systems
Abstract
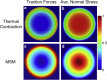
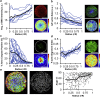
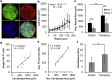
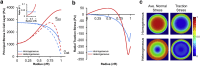
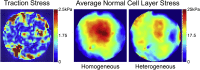
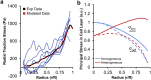
Stress fields emerging from the transfer of forces between cells within multicellular systems are increasingly being recognized as major determinants of cell fate. Current analytical and numerical models used for the calculation of stresses within cell monolayers assume homogeneous contractile and mechanical cellular properties; however, cell behavior varies by region within constrained tissues. Here, we show the impact of heterogeneous cell properties on resulting stress fields that guide cell phenotype and apoptosis. Using circular micropatterns, we measured biophysical metrics associated with cell mechanical stresses. We then computed cell-layer stress distributions using finite element contraction models and monolayer stress microscopy. In agreement with previous studies, cell spread area, alignment, and traction forces increase, whereas apoptotic activity decreases, from the center of cell layers to the edge. The distribution of these metrics clearly indicates low cell stress in central regions and high cell stress at the periphery of the patterns. However, the opposite trend is predicted by computational models when homogeneous contractile and mechanical properties are assumed. In our model, utilizing heterogeneous cell-layer contractility and elastic moduli values based on experimentally measured biophysical parameters, we calculate low cell stress in central areas and high anisotropic stresses in peripheral regions, consistent with the biometrics. These results clearly demonstrate that common assumptions of uniformity in cell contractility and stiffness break down in postconfluence confined multicellular systems. This work highlights the importance of incorporating regional variations in cell mechanical properties when estimating emergent stress fields from collective cell behavior.
Copyright © 2019 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Multicellular aligned bands disrupt global collective cell behavior.Acta Biomater. 2023 Jun;163:117-130. doi: 10.1016/j.actbio.2022.10.041. Epub 2022 Oct 26. Acta Biomater. 2023. PMID: 36306982 Free PMC article.
-
Anisotropy profoundly alters stress fields within contractile cells and cell aggregates.Biomech Model Mechanobiol. 2022 Oct;21(5):1357-1370. doi: 10.1007/s10237-022-01595-0. Epub 2022 Jul 12. Biomech Model Mechanobiol. 2022. PMID: 35829977 Free PMC article.
-
Incorporating tissue anisotropy and heterogeneity in finite element models of trabecular bone altered predicted local stress distributions.Biomech Model Mechanobiol. 2018 Apr;17(2):605-614. doi: 10.1007/s10237-017-0981-8. Epub 2017 Nov 14. Biomech Model Mechanobiol. 2018. PMID: 29139053
-
The matrix environmental and cell mechanical properties regulate cell migration and contribute to the invasive phenotype of cancer cells.Rep Prog Phys. 2019 Jun;82(6):064602. doi: 10.1088/1361-6633/ab1628. Epub 2019 Apr 4. Rep Prog Phys. 2019. PMID: 30947151 Review.
-
Physically based principles of cell adhesion mechanosensitivity in tissues.Rep Prog Phys. 2012 Nov;75(11):116601. doi: 10.1088/0034-4885/75/11/116601. Epub 2012 Oct 19. Rep Prog Phys. 2012. PMID: 23085962 Review.
Cited by
-
Mechanical Regulation of Apoptosis in the Cardiovascular System.Ann Biomed Eng. 2021 Jan;49(1):75-97. doi: 10.1007/s10439-020-02659-x. Epub 2020 Nov 9. Ann Biomed Eng. 2021. PMID: 33169343 Free PMC article. Review.
-
Force-sensing protein expression in response to cardiovascular mechanotransduction.EBioMedicine. 2024 Dec;110:105412. doi: 10.1016/j.ebiom.2024.105412. Epub 2024 Oct 30. EBioMedicine. 2024. PMID: 39481337 Free PMC article. Review.
-
Multicellular aligned bands disrupt global collective cell behavior.Acta Biomater. 2023 Jun;163:117-130. doi: 10.1016/j.actbio.2022.10.041. Epub 2022 Oct 26. Acta Biomater. 2023. PMID: 36306982 Free PMC article.
-
Spatiotemporal single-cell tracking analysis in 3D tissues to reveal heterogeneous cellular response to mechanical stimuli.Sci Adv. 2023 Oct 13;9(41):eadf9917. doi: 10.1126/sciadv.adf9917. Epub 2023 Oct 13. Sci Adv. 2023. PMID: 37831766 Free PMC article.
-
Anisotropy profoundly alters stress fields within contractile cells and cell aggregates.Biomech Model Mechanobiol. 2022 Oct;21(5):1357-1370. doi: 10.1007/s10237-022-01595-0. Epub 2022 Jul 12. Biomech Model Mechanobiol. 2022. PMID: 35829977 Free PMC article.
References
-
- Aragona M., Panciera T., Piccolo S. A mechanical checkpoint controls multicellular growth through YAP/TAZ regulation by actin-processing factors. Cell. 2013;154:1047–1059. - PubMed
-
- Lee J., Abdeen A.A., Kilian K.A. Interfacial geometry dictates cancer cell tumorigenicity. Nat. Mater. 2016;15:856–862. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

