Efficacy Of Line Probe Assay In Detection Of Drug-Resistant Pulmonary Tuberculosis In Comparison With GeneXpert And Phenotypic Methods In Iran And Genetic Analysis Of Isolates By MIRU-VNTR
- PMID: 31814746
- PMCID: PMC6863623
- DOI: 10.2147/IDR.S222905
Efficacy Of Line Probe Assay In Detection Of Drug-Resistant Pulmonary Tuberculosis In Comparison With GeneXpert And Phenotypic Methods In Iran And Genetic Analysis Of Isolates By MIRU-VNTR
Abstract
Background: Successful treatment of tuberculosis depends on early diagnosis and use of appropriate drug susceptibility testing in a timely manner. In the present study, LPA efficacy was assayed in detection and drug susceptibility testing of pulmonary tuberculosis in comparison to available methods in Iran and phylogenetic analyses of isolated cases carried out by MIRU-VNTR.
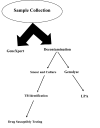
Methods: This study was conducted at the Tehran Regional Reference Laboratory for Tuberculosis. All sputum specimens were subjected to smear, culture, and drug susceptibility testing (DST), GeneXpert, and LPA. Finally, 15-locus-based MIRU-VNTR was used for molecular genotyping.
Results: From a total of 920 sputum specimens, 6.08% (n=56) were identified as MTBC by culture, 6.8% (n=63) by GeneXpert, and 6.5% (n=60) by LPA. Phenotype DST and LPA methods confirmed the resistance of 4 and 14 specimens to rifampin (RIF) and isoniazid (INH); two cases were considered as multidrug-resistant (MDR). Using GeneXpert, four cases were identified as RIF-resistant. Based on LPA results, inhA and katG mutations were detected in 100% and 21.4% of INH-resistant cases, respectively. All 56 culture positive Mycobacterium tuberculosis isolates were placed in 29 different clusters using MIRU-VNTR genotyping. Two MDR-TB, 2 RIF mono-resistant, and 12 INH mono-resistant cases were placed in different clusters.
Conclusion: LPA is an appropriate method for early detection and accurate diagnosis of TB and drug-resistant cases that makes it possible to distinguish INH mono-resistant cases from MDR cases in Iran.
Keywords: drug resistance; early diagnosis; line probe assay; mutation; tuberculosis.
© 2019 Kazemian et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures


References
-
- World Health Organization (WHO). Global tuberculosis report 2017. Available from: www.who.int/tb/publications/global_report/ Accessed October31, 2019.
-
- Kazemian H, Kardan-Yamchi J, Mosavari N, Feizabadi MM. Molecular characterization of multidrug and extensive drug-resistant Mycobacterium tuberculosis isolates from Iran. Infez Med. 2019;27(1):26–31. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

