Effectiveness and safety of the conversion to MeltDose® extended-release tacrolimus from other formulations of tacrolimus in stable kidney transplant patients: A retrospective study
- PMID: 31815310
- PMCID: PMC7050537
- DOI: 10.1111/ctr.13767
Effectiveness and safety of the conversion to MeltDose® extended-release tacrolimus from other formulations of tacrolimus in stable kidney transplant patients: A retrospective study
Abstract
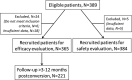
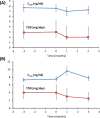
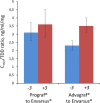
Tacrolimus is the cornerstone of immunosuppressive therapy after kidney transplantation. Its narrow therapeutic window mandates serum level strict monitoring and dose adjustments to ensure the optimal risk-benefit balance. This observational retrospective study analyzed the effectiveness and safety of conversion from twice-daily immediate-release tacrolimus (IR-Tac) or once-daily prolonged-release tacrolimus (PR-Tac) to the recent formulation once-daily MeltDose® extended-release tacrolimus (LCP-Tac) in 365 stable kidney transplant recipients. We compared kidney function three months before and three months after the conversion. Three months after conversion, the total daily dose was reduced ~35% (P < .0001), and improved bioavailability and stable serum LCP-Tac concentrations were observed. There was no increase in the number of patients requiring tacrolimus dose adjustments after conversion. Renal function was unaltered, and no cases of BPAR were reported. Reports of tremors, as collected in the clinical histories for each patient, decreased from pre-conversion (20.8%) to post-conversion (11.8%, P < .0001). LCP-Tac generated a cost reduction of 63% compared with PR-Tac. In conclusion, the conversion strategy to LCP-Tac from other tacrolimus formulations in stable kidney transplant patients showed safety and effectiveness in a real-world setting, confirming the data from RCTs. The specific pharmacokinetic properties of LCP-Tac could be potentially advantageous in patients with tacrolimus-related adverse events.
Keywords: LCPT; extended-release tacrolimus; immediate-release tacrolimus; kidney transplant; prolonged-release tacrolimus; tacrolimus.
© 2019 The Authors. Clinical Transplantation published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Allison AC. Immunosuppressive drugs: the first 50 years and a glance forward. Immunopharmacology. 2000;47(2‐3):63‐83. - PubMed
-
- Lim MA, Kohli J, Bloom RD. Immunosuppression for kidney transplantation: where are we now and where are we going? Transplant Rev (Orlando). 2017;31(1):10‐17. - PubMed
-
- Hedayat S, Kershner RP, Su G. Relationship of whole‐blood FK506 concentrations to rejection and toxicity in liver and kidney transplants. J Biopharm Stat. 1996;6(4):411‐424. - PubMed
-
- Kahan BD, Keown P, Levy GA, Johnston A. Therapeutic drug monitoring of immunosuppressant drugs in clinical practice. Clin Ther. 2002;24(3):330–350. discussion 329. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

