Chemical Synthesis of Silk-Mimetic Polymers
- PMID: 31817786
- PMCID: PMC6947416
- DOI: 10.3390/ma12244086
Chemical Synthesis of Silk-Mimetic Polymers
Abstract
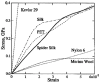
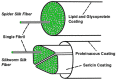
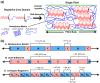
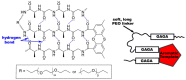
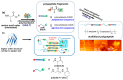
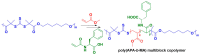
Silk is a naturally occurring high-performance material that can surpass man-made polymers in toughness and strength. The remarkable mechanical properties of silk result from the primary sequence of silk fibroin, which bears semblance to a linear segmented copolymer with alternating rigid ("crystalline") and flexible ("amorphous") blocks. Silk-mimetic polymers are therefore of great emerging interest, as they can potentially exhibit the advantageous features of natural silk while possessing synthetic flexibility as well as non-natural compositions. This review describes the relationships between primary sequence and material properties in natural silk fibroin and furthermore discusses chemical approaches towards the synthesis of silk-mimetic polymers. In particular, step-growth polymerization, controlled radical polymerization, and copolymerization with naturally derived silk fibroin are presented as strategies for synthesizing silk-mimetic polymers with varying molecular weights and degrees of sequence control. Strategies for improving macromolecular solubility during polymerization are also highlighted. Lastly, the relationships between synthetic approach, supramolecular structure, and bulk material properties are explored in this review, with the aim of providing an informative perspective on the challenges facing chemical synthesis of silk-mimetic polymers with desirable properties.
Keywords: bioinspired materials; macromolecular self-assembly; silk-mimetic polymers.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





References
-
- Hardy J.G., Römer L.M., Scheibel T.R. Polymeric materials based on silk proteins. Polymer. 2008;49:4309–4327. doi: 10.1016/j.polymer.2008.08.006. - DOI
-
- Gosline J.M., Guerette P.A., Ortlepp C.S., Savage K.N. The mechanical design of spider silks: From fibroin sequence to mechanical function. J. Exp. Biol. 1999;202:3295–3303. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

