PAK4 Kinase Activity Plays a Crucial Role in the Podosome Ring of Myeloid Cells
- PMID: 31825823
- PMCID: PMC6915307
- DOI: 10.1016/j.celrep.2019.11.016
PAK4 Kinase Activity Plays a Crucial Role in the Podosome Ring of Myeloid Cells
Erratum in
-
PAK4 Kinase Activity Plays a Crucial Role in the Podosome Ring of Myeloid Cells.Cell Rep. 2020 Apr 21;31(3):107593. doi: 10.1016/j.celrep.2020.107593. Cell Rep. 2020. PMID: 32320648 Free PMC article. No abstract available.
Abstract
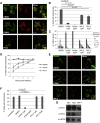
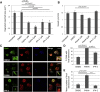
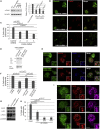
p21-Activated kinase 4 (PAK4), a serine/threonine kinase, is purported to localize to podosomes: transient adhesive structures that degrade the extracellular matrix to facilitate rapid myeloid cell migration. We find that treatment of transforming growth factor β (TGF-β)-differentiated monocytic (THP-1) cells with a PAK4-targeted inhibitor significantly reduces podosome formation and induces the formation of focal adhesions. This switch in adhesions confers a diminution of matrix degradation and reduced cell migration. Furthermore, reduced PAK4 expression causes a significant reduction in podosome number that cannot be rescued by kinase-dead PAK4, supporting a kinase-dependent role. Concomitant with PAK4 depletion, phosphorylation of Akt is perturbed, whereas a specific phospho-Akt signal is detected within the podosomes. Using superresolution analysis, we find that PAK4 specifically localizes in the podosome ring, nearer to the actin core than other ring proteins. We propose PAK4 kinase activity intersects with the Akt pathway at the podosome ring:core interface to drive regulation of macrophage podosome turnover.
Keywords: AKT; PAK4; adhesion; migration; podosome; superresolution microscopy.
Copyright © 2019 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Ahmed T., Shea K., Masters J.R., Jones G.E. A PAK4-LIMK1 pathway drives prostate cancer cell migration downstream of HGF. Cellular signalling. 2008;20:1320–1328. - PubMed
-
- Bañón-Rodríguez I., Monypenny J., Ragazzini C., Franco A., Calle Y., Jones G.E., Antón I.M. The cortactin-binding domain of WIP is essential for podosome formation and extracellular matrix degradation by murine dendritic cells. Eur. J. Cell Biol. 2011;90:213–223. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

