Toward time-resolved laser T-jump/X-ray probe spectroscopy in aqueous solutions
- PMID: 31832487
- PMCID: PMC6906120
- DOI: 10.1063/1.5129626
Toward time-resolved laser T-jump/X-ray probe spectroscopy in aqueous solutions
Abstract
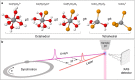
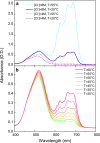
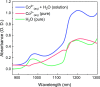
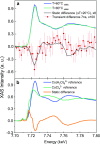
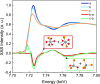
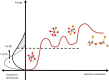
Most chemical and biochemical reactions in nature and in industrial processes are driven by thermal effects that bring the reactants above the energy barrier for reaction. In aqueous solutions, this process can also be triggered by the laser driven temperature jump (T-jump) method, in which the water vibrational (stretch, bend, or combination) modes are excited by a short laser pulse, leading to a temperature increase in the irradiated volume within a few picoseconds. The combination of the laser T-jump with X-ray spectroscopic probes would add element-specificity as well as sensitivity to the structure, the oxidation state, and the spin state of the intermediates of reactions. Here, we present preliminary results of a near infrared pump/X-ray absorption spectroscopy probe to study the ligand exchange of an octahedral aqueous Cobalt complex, which is known to pass through intermediate steps yielding tetrahedral chlorinated as final species. The structural changes of the chemical reaction are monitored with great sensitivity, even in the presence of a mild local increase in temperature. This work opens perspectives for the study of non-light-driven reactions using time-resolved X-ray spectroscopic methods.
© 2019 Author(s).
Figures
Similar articles
-
Enhancing the analysis of disorder in X-ray absorption spectra: application of deep neural networks to T-jump-X-ray probe experiments.Phys Chem Chem Phys. 2021 Apr 22;23(15):9259-9269. doi: 10.1039/d0cp06244h. Phys Chem Chem Phys. 2021. PMID: 33885072
-
Ultrafast X-ray Transient Absorption Spectroscopy of Gas-Phase Photochemical Reactions: A New Universal Probe of Photoinduced Molecular Dynamics.Acc Chem Res. 2018 Dec 18;51(12):3203-3211. doi: 10.1021/acs.accounts.8b00462. Epub 2018 Nov 21. Acc Chem Res. 2018. PMID: 30462481
-
Visualizing solution-phase reaction dynamics with time-resolved X-ray liquidography.Acc Chem Res. 2009 Feb 17;42(2):356-66. doi: 10.1021/ar800168v. Acc Chem Res. 2009. PMID: 19117426
-
The O2-Evolving Complex of Photosystem II: Recent Insights from Quantum Mechanics/Molecular Mechanics (QM/MM), Extended X-ray Absorption Fine Structure (EXAFS), and Femtosecond X-ray Crystallography Data.Acc Chem Res. 2017 Jan 17;50(1):41-48. doi: 10.1021/acs.accounts.6b00405. Epub 2016 Dec 21. Acc Chem Res. 2017. PMID: 28001034 Review.
-
Ultrafast nonlinear coherent vibrational sum-frequency spectroscopy methods to study thermal conductance of molecules at interfaces.Acc Chem Res. 2009 Sep 15;42(9):1343-51. doi: 10.1021/ar9000197. Acc Chem Res. 2009. PMID: 19388671 Review.
Cited by
-
Preface to the special issue: Selected papers from the 5th International Conference on Ultrafast Structural Dynamics.Struct Dyn. 2020 Dec 30;7(6):060402. doi: 10.1063/4.0000064. eCollection 2020 Nov. Struct Dyn. 2020. PMID: 33415181 Free PMC article. No abstract available.
-
Engraving of stainless-steel wires to improve optical quality of closed-loop wire-guided flow jet systems for optical and X-ray spectroscopy.Front Mol Biosci. 2023 Jun 14;10:1079029. doi: 10.3389/fmolb.2023.1079029. eCollection 2023. Front Mol Biosci. 2023. PMID: 37388247 Free PMC article.
-
The Role of Structural Representation in the Performance of a Deep Neural Network for X-Ray Spectroscopy.Molecules. 2020 Jun 11;25(11):2715. doi: 10.3390/molecules25112715. Molecules. 2020. PMID: 32545393 Free PMC article.
-
Nanoporous TiN/TiO2/Alumina Membrane for Photoelectrochemical Hydrogen Production from Sewage Water.Nanomaterials (Basel). 2021 Oct 6;11(10):2617. doi: 10.3390/nano11102617. Nanomaterials (Basel). 2021. PMID: 34685061 Free PMC article.
References
-
- Anslyn E. V. and Dougherty D. A., Modern Physical Organic Chemistry ( University Science Books, Sausalito, California, USA, 2006).
-
- Eigen M., “ Immeasurably fast reactions,” Nobel Lect. 11, 1963–1979 (1967).
-
- Gettins W. J. and Wyn-Jones E., “ Techniques and applications of fast reactions in solution,” in Proceedings of the NATO Advanced Study Institute on New Applications of Chemical Relaxation Spectrometry and Other Fast Reaction Methods in Solution ( Springer Science & Business Media, 1978).
LinkOut - more resources
Full Text Sources

