Distinct Laminar Requirements for NMDA Receptors in Experience-Dependent Visual Cortical Plasticity
- PMID: 31832634
- PMCID: PMC7174998
- DOI: 10.1093/cercor/bhz260
Distinct Laminar Requirements for NMDA Receptors in Experience-Dependent Visual Cortical Plasticity
Abstract
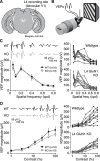
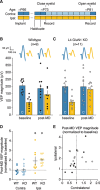
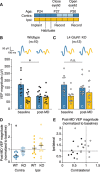
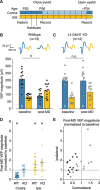
Primary visual cortex (V1) is the locus of numerous forms of experience-dependent plasticity. Restricting visual stimulation to one eye at a time has revealed that many such forms of plasticity are eye-specific, indicating that synaptic modification occurs prior to binocular integration of thalamocortical inputs. A common feature of these forms of plasticity is the requirement for NMDA receptor (NMDAR) activation in V1. We therefore hypothesized that NMDARs in cortical layer 4 (L4), which receives the densest thalamocortical input, would be necessary for all forms of NMDAR-dependent and input-specific V1 plasticity. We tested this hypothesis in awake mice using a genetic approach to selectively delete NMDARs from L4 principal cells. We found, unexpectedly, that both stimulus-selective response potentiation and potentiation of open-eye responses following monocular deprivation (MD) persist in the absence of L4 NMDARs. In contrast, MD-driven depression of deprived-eye responses was impaired in mice lacking L4 NMDARs, as was L4 long-term depression in V1 slices. Our findings reveal a crucial requirement for L4 NMDARs in visual cortical synaptic depression, and a surprisingly negligible role for them in cortical response potentiation. These results demonstrate that NMDARs within distinct cellular subpopulations support different forms of experience-dependent plasticity.
Keywords: NMDA receptor; amblyopia; long-term depression; ocular dominance plasticity; stimulus-selective response potentiation; visual cortex.
© The Author(s) 2019. Published by Oxford University Press.
Figures



Similar articles
-
Blockade of GluN2B-Containing NMDA Receptors Prevents Potentiation and Depression of Responses during Ocular Dominance Plasticity.J Neurosci. 2024 Sep 4;44(36):e0021232024. doi: 10.1523/JNEUROSCI.0021-23.2024. J Neurosci. 2024. PMID: 39117456 Free PMC article.
-
Long-term Monocular Deprivation during Juvenile Critical Period Disrupts Binocular Integration in Mouse Visual Thalamus.J Neurosci. 2020 Jan 15;40(3):585-604. doi: 10.1523/JNEUROSCI.1626-19.2019. Epub 2019 Nov 25. J Neurosci. 2020. PMID: 31767678 Free PMC article.
-
Swept contrast visual evoked potentials and their plasticity following monocular deprivation in mice.Vision Res. 2004 Dec;44(28):3381-7. doi: 10.1016/j.visres.2004.09.018. Vision Res. 2004. PMID: 15536006
-
Comparison of plasticity in vivo and in vitro in the developing visual cortex of normal and protein kinase A RIbeta-deficient mice.J Neurosci. 1998 Mar 15;18(6):2108-17. doi: 10.1523/JNEUROSCI.18-06-02108.1998. J Neurosci. 1998. PMID: 9482797 Free PMC article. Review.
-
How the mechanisms of long-term synaptic potentiation and depression serve experience-dependent plasticity in primary visual cortex.Philos Trans R Soc Lond B Biol Sci. 2013 Dec 2;369(1633):20130284. doi: 10.1098/rstb.2013.0284. Print 2014 Jan 5. Philos Trans R Soc Lond B Biol Sci. 2013. PMID: 24298166 Free PMC article. Review.
Cited by
-
Visual activity enhances neuronal excitability in thalamic relay neurons.Sci Adv. 2025 Jan 24;11(4):eadp4627. doi: 10.1126/sciadv.adp4627. Epub 2025 Jan 22. Sci Adv. 2025. PMID: 39841847 Free PMC article.
-
Silent Synapse-Based Mechanisms of Critical Period Plasticity.Front Cell Neurosci. 2020 Jul 17;14:213. doi: 10.3389/fncel.2020.00213. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32765222 Free PMC article. Review.
-
Visual Cortical Plasticity: Molecular Mechanisms as Revealed by Induction Paradigms in Rodents.Int J Mol Sci. 2023 Feb 28;24(5):4701. doi: 10.3390/ijms24054701. Int J Mol Sci. 2023. PMID: 36902131 Free PMC article. Review.
-
All-or-none disconnection of pyramidal inputs onto parvalbumin-positive interneurons gates ocular dominance plasticity.Proc Natl Acad Sci U S A. 2021 Sep 14;118(37):e2105388118. doi: 10.1073/pnas.2105388118. Proc Natl Acad Sci U S A. 2021. PMID: 34508001 Free PMC article.
-
Multiple Mechanistically Distinct Timescales of Neocortical Plasticity Occur During Habituation.Front Cell Neurosci. 2022 Apr 8;16:840057. doi: 10.3389/fncel.2022.840057. eCollection 2022. Front Cell Neurosci. 2022. PMID: 35465612 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

