Alzheimer-like amyloid and tau alterations associated with cognitive deficit in temporal lobe epilepsy
- PMID: 31834353
- PMCID: PMC6935754
- DOI: 10.1093/brain/awz381
Alzheimer-like amyloid and tau alterations associated with cognitive deficit in temporal lobe epilepsy
Erratum in
-
Erratum.Brain. 2020 Mar 1;143(3):e24. doi: 10.1093/brain/awaa007. Brain. 2020. PMID: 32333675 Free PMC article. No abstract available.
Abstract
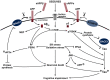
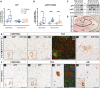
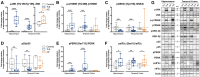
Temporal lobe epilepsy represents a major cause of drug-resistant epilepsy. Cognitive impairment is a frequent comorbidity, but the mechanisms are not fully elucidated. We hypothesized that the cognitive impairment in drug-resistant temporal lobe epilepsy could be due to perturbations of amyloid and tau signalling pathways related to activation of stress kinases, similar to those observed in Alzheimer's disease. We examined these pathways, as well as amyloid-β and tau pathologies in the hippocampus and temporal lobe cortex of drug-resistant temporal lobe epilepsy patients who underwent temporal lobe resection (n = 19), in comparison with age- and region-matched samples from neurologically normal autopsy cases (n = 22). Post-mortem temporal cortex samples from Alzheimer's disease patients (n = 9) were used as positive controls to validate many of the neurodegeneration-related antibodies. Western blot and immunohistochemical analysis of tissue from temporal lobe epilepsy cases revealed increased phosphorylation of full-length amyloid precursor protein and its associated neurotoxic cleavage product amyloid-β*56. Pathological phosphorylation of two distinct tau species was also increased in both regions, but increases in amyloid-β1-42 peptide, the main component of amyloid plaques, were restricted to the hippocampus. Furthermore, several major stress kinases involved in the development of Alzheimer's disease pathology were significantly activated in temporal lobe epilepsy brain samples, including the c-Jun N-terminal kinase and the protein kinase R-like endoplasmic reticulum kinase. In temporal lobe epilepsy cases, hippocampal levels of phosphorylated amyloid precursor protein, its pro-amyloidogenic processing enzyme beta-site amyloid precursor protein cleaving enzyme 1, and both total and hyperphosphorylated tau expression, correlated with impaired preoperative executive function. Our study suggests that neurodegenerative and stress-related processes common to those observed in Alzheimer's disease may contribute to cognitive impairment in drug-resistant temporal lobe epilepsy. In particular, we identified several stress pathways that may represent potential novel therapeutic targets.
Keywords: beta-amyloid; cognition; stress-related kinases; tau; temporal lobe epilepsy.
© The Author(s) (2019). Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures





References
-
- Aplin AE, Gibb GM, Jacobsen JS, Gallo JM, Anderton BH. In vitro phosphorylation of the cytoplasmic domain of the amyloid precursor protein by glycogen synthase kinase-3beta. J Neurochem 1996; 67: 699–707. - PubMed
-
- Appenzeller-Herzog C, Hall MN. Bidirectional crosstalk between endoplasmic reticulum stress and mTOR signaling. Trends Cell Biol 2012; 22: 274–82. - PubMed
-
- Asadi-Pooya AA, Stewart GR, Abrams DJ, Sharan A. Prevalence and incidence of drug-resistant mesial temporal lobe epilepsy in the United States. World Neurosurg 2017; 99: 662–66. - PubMed
-
- Augustinack JC, Schneider A, Mandelkow EM, Hyman BT. Specific tau phosphorylation sites correlate with severity of neuronal cytopathology in Alzheimer's disease. Acta Neuropathol 2002; 103: 26–35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

