Evolution and Diversity of Assembly-Line Polyketide Synthases
- PMID: 31838842
- PMCID: PMC6935866
- DOI: 10.1021/acs.chemrev.9b00525
Evolution and Diversity of Assembly-Line Polyketide Synthases
Abstract
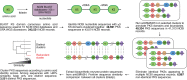
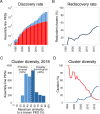
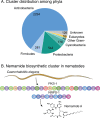
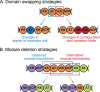
Assembly-line polyketide synthases (PKSs) are among the most complex protein machineries known in nature, responsible for the biosynthesis of numerous compounds used in the clinic. Their present-day diversity is the result of an evolutionary path that has involved the emergence of a multimodular architecture and further diversification of assembly-line PKSs. In this review, we provide an overview of previous studies that investigated PKS evolution and propose a model that challenges the currently prevailing view that gene duplication has played a major role in the emergence of multimodularity. We also analyze the ensemble of orphan PKS clusters sequenced so far to evaluate how large the entire diversity of assembly-line PKS clusters and their chemical products could be. Finally, we examine the existing techniques to access the natural PKS diversity in natural and heterologous hosts and describe approaches to further expand this diversity through engineering.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

