GCNA Preserves Genome Integrity and Fertility Across Species
- PMID: 31839537
- PMCID: PMC6946843
- DOI: 10.1016/j.devcel.2019.11.007
GCNA Preserves Genome Integrity and Fertility Across Species
Abstract
The propagation of species depends on the ability of germ cells to protect their genome from numerous exogenous and endogenous threats. While these cells employ ubiquitous repair pathways, specialized mechanisms that ensure high-fidelity replication, chromosome segregation, and repair of germ cell genomes remain incompletely understood. We identified Germ Cell Nuclear Acidic Peptidase (GCNA) as a conserved regulator of genome stability in flies, worms, zebrafish, and human germ cell tumors. GCNA contains an acidic intrinsically disordered region (IDR) and a protease-like SprT domain. In addition to chromosomal instability and replication stress, Gcna mutants accumulate DNA-protein crosslinks (DPCs). GCNA acts in parallel with the SprT domain protein Spartan. Structural analysis reveals that while the SprT domain is needed to limit DNA damage, the IDR imparts significant function. This work shows that GCNA protects germ cells from various sources of damage, providing insights into conserved mechanisms that promote genome integrity across generations.
Keywords: ACRC; DNA repair; DNA-protein crosslink; SprT domain; acidic repeat containing; chromosome instability; chromosome segregation; germ cell; intrinsically disordered region; transgenerational inheritance.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
DECLARATION OF INTERESTS
The authors declare no competing interests.
Figures


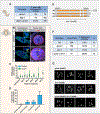

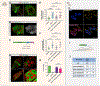
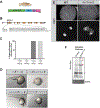

Comment in
-
When the Family Treasure Is a Doormat.Dev Cell. 2020 Jan 6;52(1):3-4. doi: 10.1016/j.devcel.2019.12.013. Dev Cell. 2020. PMID: 31951554
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 HD036247/HD/NICHD NIH HHS/United States
- P40 OD010440/OD/NIH HHS/United States
- R01 AG047318/AG/NIA NIH HHS/United States
- P01 HD078253/HD/NICHD NIH HHS/United States
- R01 GM125812/GM/NIGMS NIH HHS/United States
- P50 HD096723/HD/NICHD NIH HHS/United States
- R01 GM086647/GM/NIGMS NIH HHS/United States
- R01 GM104007/GM/NIGMS NIH HHS/United States
- R01 GM127569/GM/NIGMS NIH HHS/United States
- T32 GM008203/GM/NIGMS NIH HHS/United States
- R01 GM058800/GM/NIGMS NIH HHS/United States
- R13 CA117393/CA/NCI NIH HHS/United States
- S10 OD020103/OD/NIH HHS/United States
- P41 RR011823/RR/NCRR NIH HHS/United States
- R37 GM058800/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

