Restoration of osteochondral defects by implanting bilayered poly(lactide- co-glycolide) porous scaffolds in rabbit joints for 12 and 24 weeks
- PMID: 31844615
- PMCID: PMC6896725
- DOI: 10.1016/j.jot.2019.04.006
Restoration of osteochondral defects by implanting bilayered poly(lactide- co-glycolide) porous scaffolds in rabbit joints for 12 and 24 weeks
Abstract
Background: With the ageing of the population and the increase of sports injuries, the number of joint injuries has increased greatly. Tissue engineering or tissue regeneration is an important method to repair articular cartilage defects. While it has recently been paid much attention to use bilayered porous scaffolds to repair both cartilage and subchondral bone, it is interesting to examine to what extent a bilayer scaffold composed of the same kind of the biodegradable polymer poly(lactide-co-glycolide) (PLGA) can restore an osteochondral defect. Herein, we fabricated bilayered PLGA scaffolds and used a rabbit model to examine the efficacy of implanting the porous scaffolds with or without bone marrow mesenchymal stem cells (BMSCs). The present manuscript reports the regenerative potential up to 24 weeks.
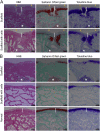
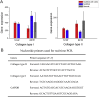
Methods: The osteochondral defect, 4 mm in diameter and 5 mm in depth, was created in the medial condyle of each knee in 23 rabbits. The bilayered PLGA scaffolds with a pore size of 100-200 μm in the chondral layer and a pore size of 300-450 μm in the osseous layer, seeded with or without BMSCs in the chondral layer, were then transplanted into the osteochondral defect of each knee. The osteochondral defect created in the same manner was untreated to act as the control. At 12 and 24 weeks postoperatively, condyles were harvested and analyzed using histology, immunohistochemistry, real-time polymerase chain reaction, and biomechanical testing to evaluate the efficacy of osteochondral repair.
Results: No joint erosion, inflammation, swelling, or deformity was observed, and all animals maintained a full range of motion. Compared with the untreated blank group, the groups implanting the bilayered scaffolds with or without cells exhibited much better resurfacing, similar to the surrounding normal tissue. The histological scores of neotissues repaired by the scaffold with cells were closer to that of normal tissue. Although the biomechanical properties of neotissues were not as good as the normal tissue, no significant difference was found between the gene levels of neotissues repaired by the scaffold with or without cells and that of the normal tissue. The repair of the osteochondral defect tends to be stable 12 weeks after implantation.
Conclusions: Our bilayered PLGA porous scaffold supports long-term osteochondral repair via in vivo tissue engineering or regeneration, and its effect can be further facilitated under the scaffold seeded with allogenic BMSCs.
The translational potential of this article: The bilayered PLGA porous scaffold can facilitate the repair of osteochondral defects and has potential for application in osteochondral tissue engineering.
Keywords: Bilayered scaffold; Osteochondral defect; Poly(lactide-co-glycolide) (PLGA); Stem cell; Tissue engineering.
© 2019 The Authors.
Figures






References
LinkOut - more resources
Full Text Sources
Research Materials

