Breast-Associated Adipocytes Secretome Induce Fatty Acid Uptake and Invasiveness in Breast Cancer Cells via CD36 Independently of Body Mass Index, Menopausal Status and Mammary Density
- PMID: 31847105
- PMCID: PMC6966437
- DOI: 10.3390/cancers11122012
Breast-Associated Adipocytes Secretome Induce Fatty Acid Uptake and Invasiveness in Breast Cancer Cells via CD36 Independently of Body Mass Index, Menopausal Status and Mammary Density
Abstract
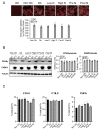
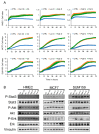
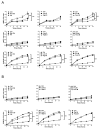
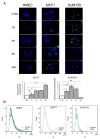
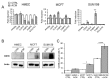
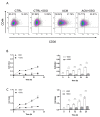
Breast adiposity is correlated with body mass index, menopausal status and mammary density. We here wish to establish how these factors influence the cross-talk between breast adipocytes and normal or malignant breast cells. Adipocyte-derived stem cells (ASCs) were obtained from healthy women and classified into six distinct groups based on body mass index, menopausal status and mammary density. The ASCs were induced to differentiate, and the influence of their conditioned media (ACM) was determined. Unexpectedly, there were no detectable differences in adipogenic differentiation and secretion between the six ASC groups, while their corresponding ACMs had no detectable influence on normal breast cells. In clear contrast, all ACMs profoundly influenced the proliferation, migration and invasiveness of malignant breast cells and increased the number of lipid droplets in their cytoplasm via increased expression of the fatty acid receptor CD36, thereby increasing fatty acid uptake. Importantly, inhibition of CD36 reduced lipid droplet accumulation and attenuated the migration and invasion of the breast cancer cells. These findings suggest that breast-associated adipocytes potentiate the invasiveness of breast cancer cells which, at least in part, is mediated by metabolic reprogramming via CD36-mediated fatty acid uptake.
Keywords: CD36; adiposity; body mass index; breast cancer; fatty acid; mammary density; menopausal status.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Adipocytes secretome from normal and tumor breast favor breast cancer invasion by metabolic reprogramming.Clin Transl Oncol. 2023 May;25(5):1389-1401. doi: 10.1007/s12094-022-03035-y. Epub 2022 Dec 15. Clin Transl Oncol. 2023. PMID: 36520383
-
Interaction with adipocyte stromal cells induces breast cancer malignancy via S100A7 upregulation in breast cancer microenvironment.Breast Cancer Res. 2017 Jun 19;19(1):70. doi: 10.1186/s13058-017-0863-0. Breast Cancer Res. 2017. PMID: 28629450 Free PMC article.
-
Adipocyte-induced CD36 expression drives ovarian cancer progression and metastasis.Oncogene. 2018 Apr;37(17):2285-2301. doi: 10.1038/s41388-017-0093-z. Epub 2018 Feb 5. Oncogene. 2018. PMID: 29398710 Free PMC article.
-
Interaction between CD36 and FABP4 modulates adipocyte-induced fatty acid import and metabolism in breast cancer.NPJ Breast Cancer. 2021 Sep 24;7(1):129. doi: 10.1038/s41523-021-00324-7. NPJ Breast Cancer. 2021. PMID: 34561446 Free PMC article.
-
Fatty acid composition and CD36 expression in breast adipose tissue of premenopausal and postmenopausal women.Anticancer Res. 1997 Mar-Apr;17(2A):1217-21. Anticancer Res. 1997. PMID: 9137475
Cited by
-
Intratumoral Adipocyte-High Breast Cancer Enrich for Metastatic and Inflammation-Related Pathways but Associated with Less Cancer Cell Proliferation.Int J Mol Sci. 2020 Aug 11;21(16):5744. doi: 10.3390/ijms21165744. Int J Mol Sci. 2020. PMID: 32796516 Free PMC article.
-
Multi‑faceted role of cancer‑associated adipocytes in the tumor microenvironment (Review).Mol Med Rep. 2021 Dec;24(6):866. doi: 10.3892/mmr.2021.12506. Epub 2021 Oct 22. Mol Med Rep. 2021. PMID: 34676881 Free PMC article. Review.
-
Fatty acid metabolism reprogramming in ccRCC: mechanisms and potential targets.Nat Rev Urol. 2023 Jan;20(1):48-60. doi: 10.1038/s41585-022-00654-6. Epub 2022 Oct 3. Nat Rev Urol. 2023. PMID: 36192502 Free PMC article. Review.
-
The metabolism of cancer cells during metastasis.Nat Rev Cancer. 2021 Mar;21(3):162-180. doi: 10.1038/s41568-020-00320-2. Epub 2021 Jan 18. Nat Rev Cancer. 2021. PMID: 33462499 Free PMC article. Review.
-
Cancer-associated adipocytes: emerging supporters in breast cancer.J Exp Clin Cancer Res. 2020 Aug 12;39(1):156. doi: 10.1186/s13046-020-01666-z. J Exp Clin Cancer Res. 2020. PMID: 32787888 Free PMC article. Review.
References
-
- Sorlie T., Perou C.M., Tibshirani R., Aas T., Geisler S., Johnsen H., Hastie T., Eisen M.B., van de Rijn M., Jeffrey S.S., et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. USA. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. - DOI - PMC - PubMed
-
- Chan D.S., Vieira A.R., Aune D., Bandera E.V., Greenwood D.C., McTiernan A., Navarro Rosenblatt D., Thune I., Vieira R., Norat T. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 2014;25:1901–1914. doi: 10.1093/annonc/mdu042. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

