Phase-Dependent Response to Afferent Stimulation During Fictive Locomotion: A Computational Modeling Study
- PMID: 31849596
- PMCID: PMC6896512
- DOI: 10.3389/fnins.2019.01288
Phase-Dependent Response to Afferent Stimulation During Fictive Locomotion: A Computational Modeling Study
Abstract
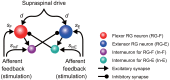
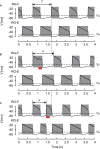
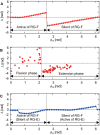
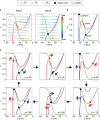
Central pattern generators (CPGs) in the spinal cord generate rhythmic neural activity and control locomotion in vertebrates. These CPGs operate under the control of sensory feedback that affects the generated locomotor pattern and adapt it to the animal's biomechanics and environment. Studies of the effects of afferent stimulation on fictive locomotion in immobilized cats have shown that brief stimulation of peripheral nerves can reset the ongoing locomotor rhythm. Depending on the phase of stimulation and the stimulated nerve, the applied stimulation can either shorten or prolong the current locomotor phase and the locomotor cycle. Here, we used a mathematical model of a half-center CPG to investigate the phase-dependent effects of brief stimulation applied to CPG on the CPG-generated locomotor oscillations. The CPG in the model consisted of two half-centers mutually inhibiting each other. The rhythmic activity in each half-center was based on a slowly inactivating, persistent sodium current. Brief stimulation was applied to CPG half-centers in different phases of the locomotor cycle to produce phase-dependent changes in CPG activity. The model reproduced several results from experiments on the effect of afferent stimulation of fictive locomotion in cats. The mechanisms of locomotor rhythm resetting under different conditions were analyzed using dynamic systems theory methods.
Keywords: afferent control of CPG; central pattern generator; dynamic structure; half-center CPG; phase-dependent response.
Copyright © 2019 Fujiki, Aoi, Tsuchiya, Danner, Rybak and Yanagihara.
Figures

Similar articles
-
Contribution of Afferent Feedback to Adaptive Hindlimb Walking in Cats: A Neuromusculoskeletal Modeling Study.Front Bioeng Biotechnol. 2022 Apr 8;10:825149. doi: 10.3389/fbioe.2022.825149. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35464733 Free PMC article.
-
Modeling the mammalian locomotor CPG: insights from mistakes and perturbations.Prog Brain Res. 2007;165:235-53. doi: 10.1016/S0079-6123(06)65015-2. Prog Brain Res. 2007. PMID: 17925250 Free PMC article. Review.
-
Chapter 2--the spinal generation of phases and cycle duration.Prog Brain Res. 2011;188:15-29. doi: 10.1016/B978-0-444-53825-3.00007-3. Prog Brain Res. 2011. PMID: 21333800 Review.
-
Flexor reflex afferents reset the step cycle during fictive locomotion in the cat.Exp Brain Res. 1998 Oct;122(3):339-50. doi: 10.1007/s002210050522. Exp Brain Res. 1998. PMID: 9808307
-
Flexor and Extensor Ankle Afferents Broadly Innervate Locomotor Spinal Shox2 Neurons and Induce Similar Effects in Neonatal Mice.Front Cell Neurosci. 2019 Oct 9;13:452. doi: 10.3389/fncel.2019.00452. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31649510 Free PMC article.
Cited by
-
Biomechanical and Sensory Feedback Regularize the Behavior of Different Locomotor Central Pattern Generators.Biomimetics (Basel). 2022 Dec 4;7(4):226. doi: 10.3390/biomimetics7040226. Biomimetics (Basel). 2022. PMID: 36546926 Free PMC article.
-
Contribution of Afferent Feedback to Adaptive Hindlimb Walking in Cats: A Neuromusculoskeletal Modeling Study.Front Bioeng Biotechnol. 2022 Apr 8;10:825149. doi: 10.3389/fbioe.2022.825149. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35464733 Free PMC article.
-
Fast and Slow Adaptations of Interlimb Coordination via Reflex and Learning During Split-Belt Treadmill Walking of a Quadruped Robot.Front Robot AI. 2021 Aug 6;8:697612. doi: 10.3389/frobt.2021.697612. eCollection 2021. Front Robot AI. 2021. PMID: 34422913 Free PMC article.
-
Contribution of Phase Resetting to Statistical Persistence in Stride Intervals: A Modeling Study.Front Neural Circuits. 2022 Jun 22;16:836121. doi: 10.3389/fncir.2022.836121. eCollection 2022. Front Neural Circuits. 2022. PMID: 35814485 Free PMC article.
-
Computational Modeling of Spinal Locomotor Circuitry in the Age of Molecular Genetics.Int J Mol Sci. 2021 Jun 25;22(13):6835. doi: 10.3390/ijms22136835. Int J Mol Sci. 2021. PMID: 34202085 Free PMC article. Review.
References
-
- Aoi S., Kondo T., Hayashi N., Yanagihara D., Aoki S., Yamaura H., et al. . (2013). Contributions of phase resetting and interlimb coordination to the adaptive control of hindlimb obstacle avoidance during locomotion in rats: a simulation study. Biol. Cybern. 107, 201–216. 10.1007/s00422-013-0546-6 - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

