2,2'-Dipyridyl diselenide: A chemoselective tool for cysteine deprotection and disulfide bond formation
- PMID: 31856422
- PMCID: PMC7509986
- DOI: 10.1002/psc.3236
2,2'-Dipyridyl diselenide: A chemoselective tool for cysteine deprotection and disulfide bond formation
Abstract
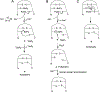
There are many examples of bioactive, disulfide-rich peptides and proteins whose biological activity relies on proper disulfide connectivity. Regioselective disulfide bond formation is a strategy for the synthesis of these bioactive peptides, but many of these methods suffer from a lack of orthogonality between pairs of protected cysteine (Cys) residues, efficiency, and high yields. Here, we show the utilization of 2,2'-dipyridyl diselenide (PySeSePy) as a chemical tool for the removal of Cys-protecting groups and regioselective formation of disulfide bonds in peptides. We found that peptides containing either Cys(Mob) or Cys(Acm) groups treated with PySeSePy in trifluoroacetic acid (TFA) (with or without triisopropylsilane (TIS) were converted to Cys-S-SePy adducts at 37 °C and various incubation times. This novel Cys-S-SePy adduct is able to be chemoselectively reduced by five-fold excess ascorbate at pH 4.5, a condition that should spare already installed peptide disulfide bonds from reduction. This chemoselective reduction by ascorbate will undoubtedly find utility in numerous biotechnological applications. We applied our new chemistry to the iodine-free synthesis of the human intestinal hormone guanylin, which contains two disulfide bonds. While we originally envisioned using ascorbate to chemoselectively reduce one of the formed Cys-S-SePy adducts to catalyze disulfide bond formation, we found that when pairs of Cys(Acm) residues were treated with PySeSePy in TFA, the second disulfide bond formed spontaneously. Spontaneous formation of the second disulfide is most likely driven by the formation of the thermodynamically favored diselenide (PySeSePy) from the two Cys-S-SePy adducts. Thus, we have developed a one-pot method for concomitant deprotection and disulfide bond formation of Cys(Acm) pairs in the presence of an existing disulfide bond.
© 2019 European Peptide Society and John Wiley & Sons, Ltd.
Figures


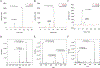




Similar articles
-
Reduction of cysteine-S-protecting groups by triisopropylsilane.J Pept Sci. 2018 Nov;24(11):e3130. doi: 10.1002/psc.3130. J Pept Sci. 2018. PMID: 30353614 Free PMC article.
-
2,2'-Bispyridyl disulfide rapidly induces intramolecular disulfide bonds in peptides.Peptides. 1999;20(7):881-4. doi: 10.1016/s0196-9781(99)00076-5. Peptides. 1999. PMID: 10477090
-
Catalytic Staudinger-Vilarrasa reaction for the direct ligation of carboxylic acids and azides.J Org Chem. 2009 Mar 6;74(5):2203-6. doi: 10.1021/jo802825e. J Org Chem. 2009. PMID: 19203231
-
[A novel synthetic approach of tryptophan-containing cystine peptides by regioselective disulfide bond-forming reaction using the silyl chloride-sulfoxide system].Yakugaku Zasshi. 2000 Feb;120(2):197-205. doi: 10.1248/yakushi1947.120.2_197. Yakugaku Zasshi. 2000. PMID: 10689966 Review. Japanese.
-
New disulfide bond-forming reactions for peptide and protein synthesis.Braz J Med Biol Res. 1994 Dec;27(12):2733-44. doi: 10.1002/chin.199620250. Braz J Med Biol Res. 1994. PMID: 7549997 Review.
Cited by
-
Techniques to Determine Mammalian Sperm Capacitation.Methods Mol Biol. 2025;2897:463-495. doi: 10.1007/978-1-0716-4406-5_31. Methods Mol Biol. 2025. PMID: 40202654
References
-
- Chang SG, Choi KD, Jang SH & Shin HC Role of disulfide bonds in the structure and activity of human insulin. Mol cells. 2003;16(3):4117–4120. - PubMed
-
- Liu F, Li P, Gelfanov V, Mayer J & DiMarchi R Synthetic advances in insulin-like peptides enable novel bioactivity. Acc. Chem. Res 2017;50(8):1855–1865. - PubMed
-
- Price-Carter M, Hull MS & Goldenberg DP Roles of individual disulfide bonds in the stability and folding of an ω-conotoxin. Biochemistry 1998;37(27):9851–9861. - PubMed
-
- Gehrmann J, Alewood PF & Craik DJ Structure determination of the three disulfide bond isomers of α-conotoxin GI: a model for the role of disulfide bonds in structural stability. J. Mol. Biol 1998;278(2):401–415. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
