Immunohistochemistry in the diagnosis and classification of neuroendocrine neoplasms: what can brown do for you?
- PMID: 31857137
- PMCID: PMC7177196
- DOI: 10.1016/j.humpath.2019.12.002
Immunohistochemistry in the diagnosis and classification of neuroendocrine neoplasms: what can brown do for you?
Abstract
This review is based on a presentation given at the Hans Popper Hepatopathology Society companion meeting at the 2019 United States and Canadian Academy of Pathology Annual Meeting. It presents updates on the diagnosis and classification of neuroendocrine neoplasms, with an emphasis on the role of immunohistochemistry. Neuroendocrine neoplasms often present in liver biopsies as metastases of occult origin. Specific topics covered include 1. general features of neuroendocrine neoplasms, 2. general neuroendocrine marker immunohistochemistry, with discussion of the emerging marker INSM1, 3. non-small cell carcinoma with (occult) neuroendocrine differentiation, 4. the WHO Classification of neuroendocrine neoplasms, with discussion of the 2019 classification of gastroenteropancreatic neoplasms, 5. use of Ki-67 immunohistochemistry, 6. immunohistochemistry to assign site of origin in neuroendocrine metastasis of occult origin, 7. immunohistochemistry to distinguish well-differentiated neuroendocrine tumor G3 from poorly differentiated neuroendocrine carcinoma, 8. lesions frequently misdiagnosed as well-differentiated neuroendocrine tumor, and 9. required and recommended data elements for biopsies and resections with associated immunohistochemical stains. Next-generation immunohistochemistry, including lineage-restricted transcription factors (e.g., CDX2, islet 1, OTP, SATB2) and protein correlates of molecular genetic events (e.g., p53, Rb), is indispensable for the accurate diagnosis and classification of these neoplasms.
Keywords: Carcinoma of Unknown Primary; Differential Diagnosis; INSM1; Immunohistochemistry; Ki-67; Neuroendocrine; World Health Organization Classification.
Copyright © 2019 Elsevier Inc. All rights reserved.
Conflict of interest statement
Nothing to Disclose: The author of this manuscript has indicated that he has no conflicts of interest that relate to the content of this manuscript.
Figures





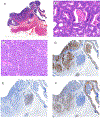






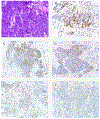


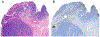


References
-
- Ordonez NG. Broad-spectrum immunohistochemical epithelial markers: a review. Hum Pathol 2013;44(7):1195–215. - PubMed
-
- Bellizzi AM. Assigning site of origin in metastatic neuroendocrine neoplasms: a clinically significant application of diagnostic immunohistochemistry. Adv Anat Pathol 2013;20(5):285–314. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

