Salinity Stress Responses and Adaptation Mechanisms in Eukaryotic Green Microalgae
- PMID: 31861232
- PMCID: PMC6952985
- DOI: 10.3390/cells8121657
Salinity Stress Responses and Adaptation Mechanisms in Eukaryotic Green Microalgae
Abstract
High salinity is a challenging environmental stress for organisms to overcome. Unicellular photosynthetic microalgae are especially vulnerable as they have to grapple not only with ionic imbalance and osmotic stress but also with the generated reactive oxygen species (ROS) interfering with photosynthesis. This review attempts to compare and contrast mechanisms that algae, particularly the eukaryotic Chlamydomonas microalgae, exhibit in order to immediately respond to harsh conditions caused by high salinity. The review also collates adaptation mechanisms of freshwater algae strains under persistent high salt conditions. Understanding both short-term and long-term algal responses to high salinity is integral to further fundamental research in algal biology and biotechnology.
Keywords: Chlamydomonas; adaptation; green algae; high salt stress; salinity; transcriptome.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
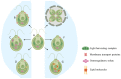


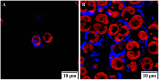

References
-
- Brock T.D. Salinity and the ecology of Dunaliella from Great Salt Lake. Microbiology. 1975;89:285–292. doi: 10.1099/00221287-89-2-285. - DOI
-
- Costelloe J.F., Powling J., Reid J.R.W., Shiel R.J., Hudson P. Algal diversity and assemblages in arid zone rivers of the Lake Eyre Basin, Australia. River Res. Appl. 2005;21:337–349. doi: 10.1002/rra.851. - DOI
-
- Hellebust J.A., Le Gresley S.M. Growth characteristics of the marine rock pool flagellate Chlamydomonas pulsatilla Wollenweber (Chlorophyta) Phycologia. 1985;24:225–229. doi: 10.2216/i0031-8884-24-2-225.1. - DOI
-
- Miyasaka H., Ohnishi Y., Akano T., Fukatsu K., Mizoguchi T., Yagi K., Isamu M., Yoshiaki I., Hiroyo M., Norio S., et al. Excretion of glycerol by the marine Chlamydomonas sp. strain W-80 in high CO2 cultures. J. Ferment. Bioeng. 1998;85:122–124. doi: 10.1016/S0922-338X(97)80367-4. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

