Neonatal Exposure to Agonists and Antagonists of Sex Steroid Receptors Affects AMH and FSH Plasma Level and Their Receptors Expression in the Adult Pig Ovary
- PMID: 31861570
- PMCID: PMC7022616
- DOI: 10.3390/ani10010012
Neonatal Exposure to Agonists and Antagonists of Sex Steroid Receptors Affects AMH and FSH Plasma Level and Their Receptors Expression in the Adult Pig Ovary
Abstract
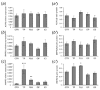
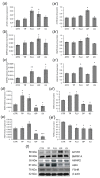
In this study piglets were injected with testosterone propionate (TP, an androgen), flutamide (FLU, an antiandrogen), 4-tert-octylphenol (OP, an estrogenic compound), ICI 182,780 (ICI, an antiestrogen) or corn oil (controls) between postnatal days 1 and 10 (N = 5/group). Then plasma anti-Müllerian hormone (AMH) and follicle stimulating hormone (FSH) concentration and the expression of their receptors were examined in the adult pig ovary. TP and FLU decreased plasma AMH and FSH concentration. In preantral follicles, TP resulted in upregulation of AMHR2 and FSHR expression, but decreased AMH protein abundance. FLU upregulated AMHR2 expression, while OP increased FSHR mRNA. In small antral follicles, OP upregulated ACVR1 and BMPR1A expression, while FLU increased BMPR1A mRNA. FLU and ICI resulted in upregulation of AMHR2 expression. TP and FLU upregulated AMH expression, while it was downregulated in response to OP or ICI. Moreover, OP and ICI resulted in downregulation of FSHR expression, while FLU decreased FSHR protein abundance. In conclusion, neonatal exposure to either agonist or antagonist of androgen receptor affected AMH and FSH signalling systems in preantral follicles. In small antral follicles these systems were influenced by compounds with estrogenic, antiestrogenic, and antiandrogenic activity. Consequently, these hormonal agents may cause an accelerated recruitment of primordial follicles and affect the cycling recruitment of small antral follicles in pigs.
Keywords: AMH; FSH; endocrine active compounds; ovary; pig.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures






Similar articles
-
Long-Term Changes in Ovarian Follicles of Gilts Exposed Neonatally to Methoxychlor: Effects on Oocyte-Derived Factors, Anti-Müllerian Hormone, Follicle-Stimulating Hormone, and Cognate Receptors.Int J Mol Sci. 2022 Mar 3;23(5):2780. doi: 10.3390/ijms23052780. Int J Mol Sci. 2022. PMID: 35269923 Free PMC article.
-
Neonatal exposure to agonists and antagonists of sex steroid receptors induces changes in the expression of oocyte-derived growth factors and their receptors in ovarian follicles in gilts.Theriogenology. 2019 Aug;134:42-52. doi: 10.1016/j.theriogenology.2019.05.018. Epub 2019 May 21. Theriogenology. 2019. PMID: 31132720
-
The impact of sex steroid agonists and antagonists on folliculogenesis in the neonatal porcine ovary via cell proliferation and apoptosis.Theriogenology. 2018 Jun;113:19-26. doi: 10.1016/j.theriogenology.2018.02.008. Epub 2018 Feb 10. Theriogenology. 2018. PMID: 29452853 Clinical Trial.
-
Interactions between androgens, FSH, anti-Müllerian hormone and estradiol during folliculogenesis in the human normal and polycystic ovary.Hum Reprod Update. 2016 Nov;22(6):709-724. doi: 10.1093/humupd/dmw027. Epub 2016 Aug 27. Hum Reprod Update. 2016. PMID: 27566840 Review.
-
[The influence of anti-Mullerian hormone on folliculogenesis].Ginekol Pol. 2008 Feb;79(2):137-40. Ginekol Pol. 2008. PMID: 18510094 Review. Polish.
Cited by
-
Long-Term Changes in Ovarian Follicles of Gilts Exposed Neonatally to Methoxychlor: Effects on Oocyte-Derived Factors, Anti-Müllerian Hormone, Follicle-Stimulating Hormone, and Cognate Receptors.Int J Mol Sci. 2022 Mar 3;23(5):2780. doi: 10.3390/ijms23052780. Int J Mol Sci. 2022. PMID: 35269923 Free PMC article.
-
Protective effect of L‑carnitine against oxidative stress injury in human ovarian granulosa cells.Exp Ther Med. 2023 Feb 23;25(4):161. doi: 10.3892/etm.2023.11860. eCollection 2023 Apr. Exp Ther Med. 2023. PMID: 36936706 Free PMC article.
-
How does chronic unpredictable mild stress affect the number of mature oocytes? An experimental study.Int J Reprod Biomed. 2025 Jun 10;23(4):303-312. doi: 10.18502/ijrm.v23i4.18782. eCollection 2025 Apr. Int J Reprod Biomed. 2025. PMID: 40766855 Free PMC article.
References
-
- Durlinger A.L., Gruijters M.J., Kramer P., Karels B., Ingraham H.A., Nachtigal M.W., Uilenbroek J.T., Grootegoed J.A., Themmen A.P. Anti-Müllerian hormone inhibits initiation of primordial follicle growth in the mouse ovary. Endocrinology. 2002;143:1076–1084. doi: 10.1210/endo.143.3.8691. - DOI - PubMed
-
- Durlinger A.L., Gruijters M.J., Kramer P., Karels B., Kumar T.R., Matzuk M.M., Rose U.M., de Jong F.H., Uilenbroek J.T., Grootegoed J.A., et al. Anti-Müllerian hormone attenuates the effects of FSH on follicle development in the mouse ovary. Endocrinology. 2001;142:4891–4899. doi: 10.1210/endo.142.11.8486. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

