Chaperoning STAT3/5 by Heat Shock Proteins: Interest of Their Targeting in Cancer Therapy
- PMID: 31861612
- PMCID: PMC7017265
- DOI: 10.3390/cancers12010021
Chaperoning STAT3/5 by Heat Shock Proteins: Interest of Their Targeting in Cancer Therapy
Abstract
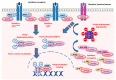
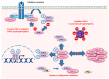
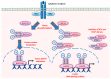
While cells from multicellular organisms are dependent upon exogenous signals for their survival, growth, and proliferation, commitment to a specific cell fate requires the correct folding and maturation of proteins, as well as the degradation of misfolded or aggregated proteins within the cell. This general control of protein quality involves the expression and the activity of molecular chaperones such as heat shock proteins (HSPs). HSPs, through their interaction with the STAT3/STAT5 transcription factor pathway, can be crucial both for the tumorigenic properties of cancer cells (cell proliferation, survival) and for the microenvironmental immune cell compartment (differentiation, activation, cytokine secretion) that contributes to immunosuppression, which, in turn, potentially promotes tumor progression. Understanding the contribution of chaperones such as HSP27, HSP70, HSP90, and HSP110 to the STAT3/5 signaling pathway has raised the possibility of targeting such HSPs to specifically restrain STAT3/5 oncogenic functions. In this review, we present how HSPs control STAT3 and STAT5 activation, and vice versa, how the STAT signaling pathways modulate HSP expression. We also discuss whether targeting HSPs is a valid therapeutic option and which HSP would be the best candidate for such a strategy.
Keywords: chaperones; heat shock proteins; stabilization; targeted therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
HSPs/STAT3 Interplay Sustains DDR and Promotes Cytokine Release by Primary Effusion Lymphoma Cells.Int J Mol Sci. 2023 Feb 15;24(4):3933. doi: 10.3390/ijms24043933. Int J Mol Sci. 2023. PMID: 36835344 Free PMC article.
-
Heat Shock Proteins and PD-1/PD-L1 as Potential Therapeutic Targets in Myeloproliferative Neoplasms.Cancers (Basel). 2020 Sep 11;12(9):2592. doi: 10.3390/cancers12092592. Cancers (Basel). 2020. PMID: 32932806 Free PMC article. Review.
-
Targeting heat shock proteins in cancer.Cancer Lett. 2013 May 28;332(2):275-85. doi: 10.1016/j.canlet.2010.10.014. Epub 2010 Nov 13. Cancer Lett. 2013. PMID: 21078542 Review.
-
Chaperoning the Cancer: The Proteostatic Functions of the Heat Shock Proteins in Cancer.Recent Pat Anticancer Drug Discov. 2017;12(1):35-47. doi: 10.2174/1574892811666161102125252. Recent Pat Anticancer Drug Discov. 2017. PMID: 27809750 Review.
-
Heat-Shock Proteins in Leukemia and Lymphoma: Multitargets for Innovative Therapeutic Approaches.Cancers (Basel). 2023 Feb 3;15(3):984. doi: 10.3390/cancers15030984. Cancers (Basel). 2023. PMID: 36765939 Free PMC article. Review.
Cited by
-
HSPs/STAT3 Interplay Sustains DDR and Promotes Cytokine Release by Primary Effusion Lymphoma Cells.Int J Mol Sci. 2023 Feb 15;24(4):3933. doi: 10.3390/ijms24043933. Int J Mol Sci. 2023. PMID: 36835344 Free PMC article.
-
Targeting STAT3 and STAT5 in Cancer.Cancers (Basel). 2020 Jul 22;12(8):2002. doi: 10.3390/cancers12082002. Cancers (Basel). 2020. PMID: 32707820 Free PMC article.
-
HSP110 Inhibition in Primary Effusion Lymphoma Cells: One Molecule, Many Pro-Survival Targets.Cancers (Basel). 2023 Nov 29;15(23):5651. doi: 10.3390/cancers15235651. Cancers (Basel). 2023. PMID: 38067355 Free PMC article.
-
Small Molecule Screening Identifies HSP90 as a Modifier of RNA Foci in Myotonic Dystrophy Type 1.Mol Cell Biol. 2025;45(6):225-237. doi: 10.1080/10985549.2024.2408025. Epub 2024 Oct 17. Mol Cell Biol. 2025. PMID: 39415708
-
Mild heat treatment in vitro potentiates human adipose stem cells: delayed aging and improved quality for long term culture.Biomater Res. 2023 Nov 27;27(1):122. doi: 10.1186/s40824-023-00448-w. Biomater Res. 2023. PMID: 38008757 Free PMC article.
References
-
- Welch W.J. Heat shock proteins functioning as molecular chaperones: Their roles in normal and stressed cells. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1993;339:327–333. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

